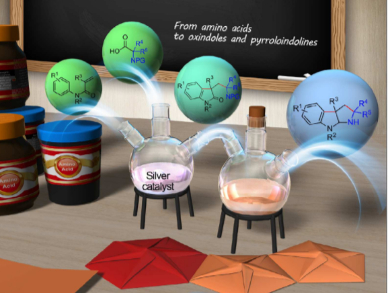
Amino acids are useful synthetic precursors for the synthesis of biologically active drugs as well as natural products due to their availability, structural diversity, and stability. Amino acids are also widely used in synthetic organic chemistry as organocatalysts and ligands to tune reactivity and control selectivity.
Kyalo Stephen Kanyiva, Sohei Makino, and Takanori Shibata, Waseda University, Japan, have developed a decarboxylative silver-catalyzed protocol that enables the coupling of an α-aminoalkyl moiety derived from α-amino acids with N-arylacrylamides to afford oxindoles. The team used AgNO3 as the catalyst and a benzyl protecting group on the N-arylacrylamide. The reaction tolerates a wide range of functional groups and provides access to structurally diverse oxindoles in moderate to high yields.
Importantly, the reaction is carried out in aqueous media and does not require rigorous removal of air or oxygen. The researchers have also demonstrated that the obtained oxindoles could be easily converted into densely functionalized pyrroloindolines by deprotection and cyclization in one pot. Both oxindoles and pyrroloindolines are prevalent in biologically active molecules and are useful synthetic intermediates.
- Silver-Catalyzed Efficient Synthesis of Oxindoles and Pyrroloindolines via α-Aminoalkylation of N-Arylacrylamides with Amino Acid Derivatives,
Kyalo Stephen Kanyiva, Sohei Makino, Takanori Shibata,
Chem. Asian J. 2018.
https://doi.org/10.1002/asia.201701739