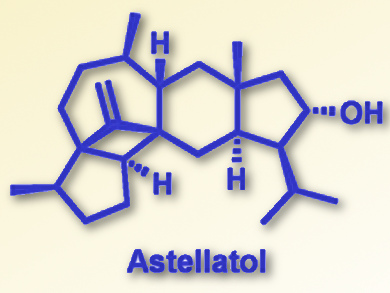
Nearly 30 years after its first structural elucidation, Chinese researchers have finally accomplished the total synthesis of astellatol, which is one of the most challenging members of the sesterterpene family of natural compounds. Their retrosynthetic approach comprises 25 steps to construct the multiring scaffold. Synthetic highlights are an unprecedented reductive radical addition to forge a congested cyclobutane and a strategic oxidation/hydrogenation step to give the substituted hydrindane moiety.
Toward the Sesterterpenes
The sesterterpenes are a rare class of natural compounds produced by fungi, lichens, marine organisms, and insects. They are associated with diverse biological properties such as anti-inflammatory, cytotoxic, anticancer, antimicrobial, and anti-biofilm activities. Nature’s biosynthesis of a sesterterpene starts with the lipid geranylfarnesol pyrophosphate. It folds, reconnects, and reorganizes its 25-carbon chain, and then, it ends up with a multiply substituted hydrocarbon ring system containing very few carbon-carbon double bonds, one or more oxo functionalities, and one isopropyl or isopropenyl moiety. Of course, the total synthesis of such an architecture is a challenge.
In 1985, E. J. Corey succeeded with retigeranic acid B [1], a sesterterpene with hydrindane motif, using a retrosynthetic approach. Astellatol, which is also a sesterterpene with hydrindane scaffold and was originally isolated from an Aspergillus variety, has an even more challenging structure. Now, it has been reconstituted by Jing Xu, Southern University of Science and Technology and Shenzhen Grubbs Institute, China, and colleagues.
Retrosynthetic Approach
In astellatol, the hydrindane moiety, which is a cyclohexane–cyclopentane ring system, is fused to a bicycle of cyclooctane and cyclobutane that in turn is connected to a substituted cyclopentane unit. In total, the synthetic chemist faces the task to construct not only the highly substituted trans-hydrindane motif, but also ten stereocenters, three of which are all-carbon, a cyclobutane, and an exo-methylene group. Several retrosynthetic approaches have been developed over the years, but none proved successful. Herein, Xu and his co-workers took advantage of a recently developed method to construct the hydrindane skeleton relatively quickly through an intramolecular reaction of an alkyne with two alkenes. This would be a good starting point for this part of the molecule.
All other procedures were taken from the established synthesis toolbox. Among their key strategies, the researchers especially highlighted a finely tuned reductive radical addition using samarium iodide and a homogenous face-selective hydrogenation. In total, the scientists converted a small starting molecule, which was derived from pulegone, a natural product frequently used in asymmetric synthesis, into the complicated architecture of the sesterterpene, using 25 steps and ending with less than one percent overall yield—which is not bad, as the researchers emphasize: except for the very last stages, all intermediate products were produced in multigram to gram scale.
New Synthetic Tools
Nature seeks to produce chemical weapons, but scientists want to learn. Besides accomplishing a challenging total synthesis, the researchers established new methods, one is closing a congested cyclobutane ring, the other furnishing the substituted hydrindane stereoisomer. Based on these experiences, new terpenoid synthetic puzzles are envisaged.
- Total Synthesis of Astellatol,
Nan Zhao, Shuqiang Yin, Shengling Xie, Hao Yan, Pan Ren, Gui Chen, Fang Chen, Jing Xu,
Angew. Chem. Int. Ed. 2018.
https://doi.org/10.1002/anie.201800167
References
- [1] Total synthesis of (±)-retigeranic acid,
E. J. Corey, Manoj C. Desai, Thomas A. Engler,
J. Am. Chem. Soc. 1985, 107, 4339–4341.
https://doi.org/10.1021/ja00300a049