Squalene, a ubiquitous unsaturated hydrocarbon originally obtained from shark liver oil, is an important precursor for the synthesis of cholesterol and steroid hormones. The cyclization mechanisms of squalene by triterpene cyclases are complex and have, therefore, attracted much attention during the past 75 years.
Tsutomu Hoshino and colleagues, Niigata University, Japan, have studied squalene-hopene cyclase (SHC) for the past 25 years. SHC converts the flexible squalene molecule into a 6,6,6,6,5-fused pentacyclic hopane scaffold as a result of the folding of squalene into an all chair conformation. SHC is found in many bacteria but is most readily isolated from the thermophilic bacterium Alicyclobacillus acidocaldarius. Ala306 residue in A. acidocaldarius SHC is widely found in known SHCs.
The steric bulk at Ala306 has a critical role in determining the stereochemical fate during the polycyclization reaction. Increases in the steric bulk at Ala306 (A306T and A306V variants) led to the accumulation of tetracyclic compounds with 20R-configuration into 6,6,6,5-fused tetracyclic scaffolds. This indicates that squalene was folded in a chair-chair-chair-boat conformation instead an all chair confirmation as described above.
In addition, SHC cyclizes the oxidized variant of squalene, 2,3-oxidosqualenes, to form 3-hydroxyhopane skeletons through polycyclization initiated at the epoxide ring. This work is the first to report the polycyclization of oxidosqualene starting from the terminal double bond (pictured below).
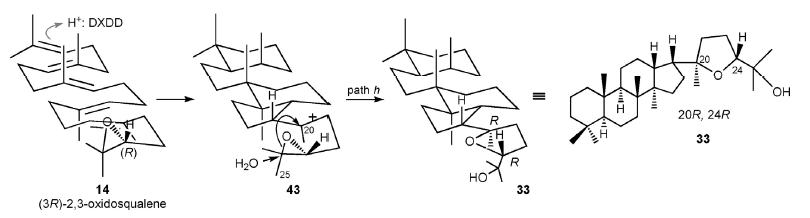
- Alicyclobacillus acidocaldarius Squalene-Hopene Cyclase: The Critical Role of Steric Bulk at Ala306 and the First Enzymatic Synthesis of Epoxydammarane from 2,3-Oxidosqualene,
Natsumi Ideno, Shikou Umeyama, Takashi Watanabe, Mami Nakajima, Tsutomu Sato, Tsutomu Hoshino,
ChemBioChem 2018.
https://doi.org/10.1002/cbic.201800281