All the Pros and None of the Cons of Different Catalysts
Heterogeneous catalysis with all its pros and cons relies heavily on porous metal oxide materials as supports. These materials provide the huge surface-to-volume ratio necessary for the deposition of an active phase that then, in turn, offers up the requisite surface area to the reagents. However, the identification of zeolites, many years ago, as functional catalysts rather than as mere supporting materials, has opened up a new perspective on porous materials.
In this context, Chathakudath P. Vinod, CSIR National Chemical Laboratory, Pune, India, and Academy of Scientific and Innovative Research (AcSIR), New Delhi, India, and colleagues have looked at aluminosilicate spheres with a macroporous core and a mesoporous shell. They have demonstrated the great utility of this proof-of-principle reagent in an archetypal C–N bond formation reaction, namely, direct N-alkylation. The researchers used mesoporous shell@macroporous core aluminosilicates to demonstrate the potential of this approach.
Solid Approach to Green Chemistry
The team explains how they used a sol-gel method with cetyltrimethyl ammonium bromide (CTAB) as the structure-directing agent in a basic medium. The process results in the selective incorporation of aluminum atoms into the silica matrix to form a relatively acidic aluminosilicate with a vast internal network of interconnected pores.
Such solid acids can, the team suggests, readily replace their homogeneous counterparts, providing more of the benefits of this catalytic power without the complex separation and regeneration cycles needed with homogeneous catalysts. The team adds that replacing the homogeneous catalyst with a solid acid could also reduce waste and reagent use, as well as improve efficiency and, thus, make such reactions “greener”.
Safer and More Environmentally Friendly
At least one step in the conventional reactions usually involves an inconvenient hydrogenation step with an expensive metal catalyst, such as ruthenium, palladium, or nickel, at high pressure. Avoiding those two factors will reduce the overall environmental impact considerably.
Even though a strategy known as “hydrogen borrowing chemistry” that uses one of the starting materials or even the solvent itself as a hydrogen source has been developed, that approach still requires metal catalysts, specific ligands/metal complexes, and harsh reaction conditions and generates what the researchers describe as “copious amounts of environmentally hazardous by-products”. Avoiding high-pressure vessels also reduces the safety concerns of the reaction scheme. The team’s mesoporous shell@macroporous core aluminosilicates could also, therefore, improve the commercial and practical viability of reactions that might otherwise be avoided in favor of multi-step approaches.
Tuning Up
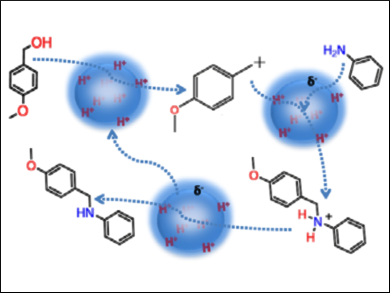
With the developed approach, there is the added benefit of the solid acid’s activity and specificity being tunable. Indeed, the researchers have selected 4-methoxy benzyl alcohol and aniline as the starting materials and tuned their solid acid accordingly. “From the point of view of the starting materials and products, the reaction resembles that of hydrogen borrowing chemistry”, they explain, “but the presence of acidity leads the reaction down an alternate pathway where Brønsted acidity catalyzes the reaction”.
The researchers obtain excellent yields using their nanocatalyst, which they named MMAS-40. They describe their approach as being a viable “green route for the synthesis of secondary amines by the cross-coupling of anilines with aromatic alcohols”. They add that the approach should be quite general for poorly nucleophilic anilines.
“The next step is to control the size of these meso-macro spheres below 100 nm and to achieve monodispersity, whereby better activity should be achieved”, Prabhakaran told ChemViews Magazine.
- Mesoporous shell@macroporous core aluminosilicates as sustainable nanocatalysts for direct N-alkylation of amines,
A. C. Sunil Sekhar, Anumol Erumpukuthickal Ashokkumar, C. T. Cygnet, S. Vidhya Lakshmi, F. L. Deepak, Chathakudath P. Vinod,
ChemNanoMat 2018.
https://doi.org/10.1002/cnma.201800081