Persistent Reordered Structures
Stimuli-sensitive materials can respond to physical forces with structural phase transitions. This also applies to biopolymer–surfactant mixtures, according to a study by Kai Liu, Changchun Institute of Applied Chemistry, Chinese Academy of Sciences, Andreas Herrmann, Zernike Institute for Advanced Materials, Groningen, The Netherlands, and RWTH Aachen University, Germany, and colleagues. Surprisingly, the newly adopted phases persist after removal of the stress and can be detected by a simple optical read-out technology. Biometric fingerprint detection is an attractive application for this technology.
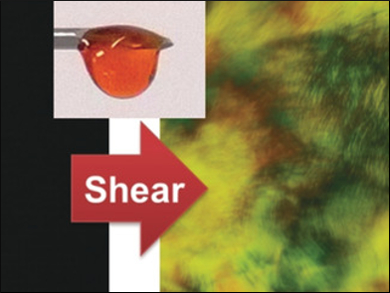
Liquid crystals are shape-anisotropic molecules that can adopt distinct ordered phases, depending on the physical conditions. Temperature, pressure, or charge can produce color shifts, dark–light switches, or a birefractive appearance, all of which represent changes in the molecular order. Such transitions can also occur in gels, and even in soaps with micellar transitions. The chemical system developed by the researchers is a complex of a supercharged polypeptide with a cationic surfactant. The viscous liquid adopted birefringence patterns after simply being touched, to reveal details such as those of a fingerprint.
Supercharged Polypeptide Fluids
Seeking to explore the behavior of biological fluids, the scientists designed a series of supercharged polypeptides that form biological soft materials with interesting properties when paired with molecules supplying the opposite charge. The supercharged polypeptides consisted of five amino acid repeating units with one or two negatively charged glutamic acid residues within each unit.
As the cationic surfactant, the researchers designed an aromatic azobenzene with a positive charge on one side and a hydrophobic chain on the other. Added together, the polypeptide and the surfactant formed a water-rich polypeptide liquid droplet with an orange hue. In this liquid, the scientists found no molecular order, birefringence, or diffraction pattern, and merely an isotropic viscous fluid.
Birefringence Read-Out
A shear force stimulated a different response. Flowing water or the touch of a finger made the sample birefringent, and ordered patterns were evident, the team reported. These ordered structures resembled the long-range lyotropic liquid crystalline phases typical for surfactant-containing mixtures. Surprisingly, that order persisted, even after removing the shear. A polarized optical microscope detected birefringence patterns that sensitively recorded the texture of the shear-applying tool. In other words, the minutiae, the ridges and lines on the fingertip that make up a fingerprint, were well-represented in the polarization micrographs.
This remarkable discovery suggests that the supercharged polypeptide fluid could, in principle, be used for biometric detection. Whereas modern-day fingerprint sensors that are not based on ink printing rely on finely adjusted electronics, the scientists present a different setup with microscopic birefringence read-out. However, the exact conditions for the phase transitions in the material and the underlying mechanisms have yet to be explored, the researchers remark.
- Genetically Engineered Supercharged Polypeptide Fluids: Fast and Persistent Self-Ordering Induced by Touch,
Lei Zhang, Chao Ma, Jing Sun, Baiqi Shao, Giuseppe Portale, Dong Chen, Kai Liu, Andreas Herrmann,
Angew. Chem. Int. Ed. 2018.
https://doi.org/10.1002/anie.201803169