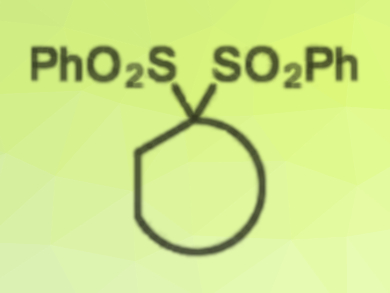
Bis(sulfones) (pictured) are often used as linker groups in cyclization reactions because they simplify substrate synthesis. Once they have served this purpose, the sulfones are typically no longer wanted because they have limited utility for future steps.
Barry Trost and Christopher Kalnmals, Stanford University, CA, USA, have reported a transition‐metal‐free coupling process whereby bis(sulfones) combine with organolithium reagents to generate trisubstituted alkenes. It showss that, although bis(sulfones) usually act as sp3 nucleophiles, they can also behave as sp2 electrophiles when treated with organolithium reagents. In this transformation, the alkyl- or aryllithium acts as both a nucleophile and a base. The first equivalent of lithium reagent promotes an E2 elimination of a sulfinate anion to form a vinyl sulfone. The second equivalent adds across this vinyl sulfone, which eliminates sulfinate again to generate a trisubstituted alkene.
These reactions are nearly instantaneous at –78 °C and tolerate functional groups such as aryl and vinyl bromides, which normally react with organolithiums. This transition metal-free coupling shows that bis(sulfones) are useful synthetic tools that can be introduced as sp3 nucleophiles and then transformed into vinyl cation equivalents. This polarity inversion (“umpolung”) strategy enhances the utility of this common linker group.
- Sulfones as Synthetic Linchpins: Transition-Metal-Free sp3–sp2 and sp2–sp2 Cross-Couplings Between Geminal Bis(sulfones) and Organolithium Compounds,
Barry M. Trost, Christopher A. Kalnmals,
Chem. Eur. J. 2018.
https://doi.org/10.1002/chem.201800118