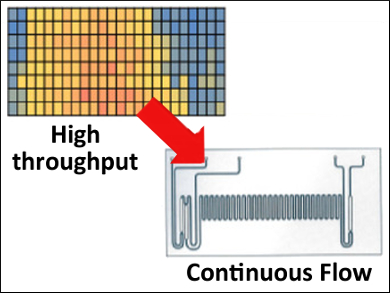
Traditional methods to discover optimal reaction conditions for small molecule synthesis is a time‐consuming effort that requires large quantities of labor and materials. High‐throughput techniques are used to screen reaction conditions. Automated reactions can be run in parallel, but the downstream analysis is typically a bottleneck due to relatively slow chromatographic separation and/or quantitation methods. High‐throughput techniques coupled with rapid mass spectrometry (MS) analysis can speed up both the discovery and optimization of reaction conditions, particularly in the cases of chemical process development and (bio)pharmaceutical drug development where pressures to shorten the time to market are increasing. However, the methode has been challenging in the case of organic reaction optimization, especially for catalytic reactions that may use solid catalysts or volatile organic solvents.
David H. Thompson and colleagues, Purdue University, West Lafayette, IN, USA, and colleagues have described a robotic high‐throughput technique (to carry out reactions in multi‐well plates) that is coupled with continuous reaction monitoring using electrospray ionization mass spectrometry (ESI‐MS) (to speed up the reaction screening process). Palladium‐catalyzed Suzuki–Miyaura (S-M) cross‐coupling reactions were screened in this system. Reaction heat maps were generated to identify the best conditions for subsequent scale-up under continuous flow conditions. The S‐M reaction was chosen because carbon‐carbon bond formation via palladium‐catalyzed S‐M cross‐coupling is an important reaction for small molecule synthesis.
The robotic high‐throughput screening reactions were done in 96‐well plates for 1 h at 50 °C, 100 °C, 150 °C, and 200 °C. The reactions were quenched by diluting the mixtures into 384‐well plates. Then they were analyzed with a triple quadrupole mass spectrometer coupled with an autosampler. A total of 648 unique experiments using 4-hydroxyphenylboronic acid and 11 different aryl halides were analyzed. The cross‐coupling reactions were evaluated as a function of stoichiometry, temperature, concentration, order of addition, and substrate type. Positive reaction conditions were identified and failed reaction conditions were fast eliminated.
The high‐throughput technique was validated using quantitative analyses of microfluidic reactions by HPLC/MS-MS. According to the researchers, their approach could also be used to optimize other catalytic and noncatalytic reactions and it could help discover highly efficient routes to pharmaceutically important molecules.
- High Throughput Experimentation and Continuous Flow Validation of Suzuki-Miyaura Cross-Coupling Reactions,
Zinia Jaman, Ahmed Mufti, Samyukta Sah, Larisa Avramova, David H. Thompson,
Chem. Eur. J. 2018.
https://doi.org/10.1002/chem.201801165