Charge separation in solid-state materials is essential for the development of photo- and electroactive systems such as solid photocatalysts and photovoltaic cells. The key to designing materials that are capable of charge separation is the spatial arrangement of electron donors and acceptors.
Norihiro Mizoshita and Shinji Inagaki, Toyota Central R&D Laboratories, Inc., Nagakute, Japan, have developed mesoporous materials with a photoinduced charge separation in the pore walls. The materials are based on a periodic mesoporous organosilica (PMO), with a framework made of hydrogen-bonded organosilica columns (pictured above). Three electroactive organic compounds with different electron‐donating and electron‐accepting properties, i.e., naphthalene, naphthalimide, and viologen derivatives, were selectively bound to the external surface of the particle, the inside of the pore wall, or the surface of the internal mesopores to control the direction of charge transfer (pictured below).
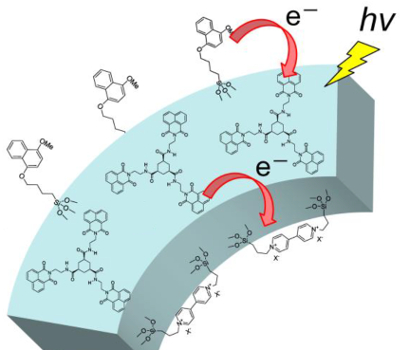
The irradiation of the resulting multifunctionalized mesoporous materials results in a charge transfer from the external surface to the pore wall and from the pore wall to the surface of the internal mesopores. The electron transfer was detected using measurements of the material’s fluorescence properties. According to the researchers, this type of functionalization of PMOs could be an effective approach to the construction of solid-state photocatalysts.
- Charge Separation in Multifunctionalized Framework of Hydrogen-Bonded Periodic Mesoporous Organosilica,
Norihiro Mizoshita, Shinji Inagaki,
Chem. Asian J. 2018.
https://doi.org/10.1002/asia.201800779