The EuChemS Chemistry Congress is the largest European chemistry congress. It is held every other year in a different European country. Events so far have taken place in Budapest, Torino, Nuremberg, Prague, Istanbul, Seville, and now in Liverpool, hosted by the Royal Society of Chemistry (RSC).
“Chemistry has been our past, is our present, and will certainly be our future.” Professor Carol Robinson, President of the RSC from the University of Oxford, UK, Professor Tom Brown, Co-chair of the scientific committee and chair of the Local organizing committee from the University of Oxford, UK, and Professor David Cole-Hamilton, Vice-President of EuChemS from the University of St. Andrews, UK, welcomed the approx. 1,400 participants from around the world to the 7th EuChemS Chemistry Congress (7ECC) held at the Liverpool Convention Center, UK.
Opening Ceremony
The five-day event brought together researchers from across the chemical sciences. Science is a global endeavor, collaborations and discussions are central. Therefore, the EuChemS Chemistry Congress represents the crossroads of many developing fields and, as Professor Bengt Nordén, Scientific Committee Chair from the Chalmers University of Technology, Sweden, said: “It is my hope that it may inspire interdisciplinary collaborations of scientists in different branches”. The wide selection of topics ranged from biological catalysis and new approaches to clean fuels to smart polymers and molecular machines.
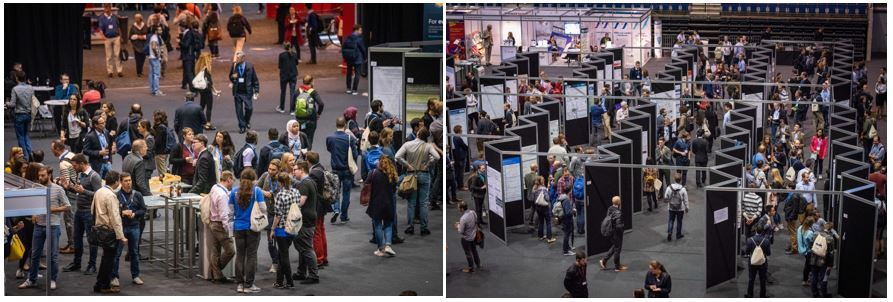
Besides lectures, posters, and panel discussions, a wealth of activities was offered, including workshops covering social and policy issues, general interest lectures, a program for early career chemists prepared by the European Young Chemists’ Network (EYCN), many networking opportunities, a communication training session, and the presentation of the historical landmark program of EuChemS. The latter aims to reinforce the sense of belonging of European chemists and to emphasize that chemistry has played a vital role in the history of Europe.
EuCheMS, originally the European Association for Chemical and Molecular Sciences, is now spelled EuChemS and has been renamed to European Chemical Society. This, as Professor Pilar Goya Laza, President of EuChemS from the Instituto de Química Médica (IQM), Madrid, Spain, explained, expresses the role of the association coordinating the efforts of the more than 40 European member chemical societies much better.
Professor Francesco De Angelis, University of l’Aquila, Italy, and Honorary Fellow of ChemPubSoc Europe, Professor Sergio Facchetti, Milan, Italy, and Professor Reiner Salzer [1], University of Dresden, Germany, were awarded the EuChemS Award for Service 2018. The award recognizes the outstanding commitment in fostering chemistry in Europe along with the activities and goals of EuChemS.
Selected Talks
The scientific part of the 7th EuChemS Chemistry Congress in Liverpool started off with a plenary lecture by Professor Ben Feringa, University of Groningen, The Netherlands, who shared the 2016 Nobel Prize in chemistry with Jean-Pierre Sauvage and J. Fraser Stoddart. In fact, it was not just the opening lecture but the debut for the European Chemistry Gold Medal award lecture. As Herman Overkleeft, Leiden University, The Netherlands, chair of the EuChemS Chemistry Gold Medal committee, explained in the introductory remarks, “nominees should be scientists who have made outstanding contributions to the field of chemistry and have significantly advanced our understanding of a particular discipline”—criteria that Feringa met without any doubt.
Figure 1. Ben Feringa is the first winner of the European Chemistry Gold Medal. Pictures: © Royal Society of Chemistry/MPP.
|
Light Creates Motion Within Single Molecules He began the lecture with a reference to the Wright brothers’ first flights at Kitty Hawk in 1903, showing a picture of their self-built flying machine. Mankind has always been fascinated by flying birds, however, what the Wright brothers came up with was not a copy or mimic of the way birds flap their wings but a completely new approach. As Feringa pointed out, a hundred years after the Wright brothers we still cannot make an artificial pigeon, “but we are extremely good in making molecules”. Yet, we are not very good at making something that is dynamic, e.g., the ribosome. But what chemists have already achieved, in his lab, in particular, was demonstrated in a lucid way in his lecture. The beginnings can be dated to the early 1990’s when molecular switches—binary systems at the molecular level—were developed by Feringa and his team. At that time control of chirality and directionality could already be addressed. Later molecular rotors—systems that, in contrast to molecular switches, can go back and forth—could be developed and Feringa explained how flexibility, viscosity, and molecular interactions need to be considered rather than gravity, which is more important when developing a car on a macroscopic scale. One of the most fascinating parts of the lecture was the presentation of a nanocar, a system that is built on molecular rotors with the added challenge of controlling and synchronizing motions to obtain a four-wheeled propulsion system. It took the team years to develop it. More recently, Feringa became interested in photopharmacology, i.e., the idea of switching medicine on with a light signal. In his lecture, he elaborated on newly developed photocontrolled antibiotics that are switchable with red light. Ben Feringa not only gives talks to scientists, as he did at the 7th EuCheMS Chemistry Congress, but occasionally, he also addresses audiences of laypersons and even children at elementary schools. From them, too, Feringa can draw inspiration when they ask him questions such as “do you have traffic jams with your nanocars?” In addition to being mesmerized by the cutting-edge research on molecular machines, as always in a talk by Ben Feringa, the audience could feel his enthusiasm and dedication. A perfect start for the conference! |
Linking Molecular Building Blocks by Strong Bonds to Make Crystalline Extended Structures
One of the plenary talks was held by Professor Omar Yaghi, University of California, Berkeley, USA, who talked about the enormous potential of metal-organic frameworks, which was uncovered within the last 20 years. Metal-organic frameworks (MOFs), also called porous coordination polymers, are a class of crystalline solid materials. Their structure consists of a regular, two- or three-dimensional network of metal cations or metal oxide clusters connected by organic molecules, also called linkers.
At the beginning of his talk, Omar Yaghi briskly walked through history, starting in 1916 with Gilbert N. Lewis who outlined how atoms can be linked together to make molecules [2] and moving on to 1959, when the first metal-organic coordination network was reported [3]. The crystal consisted of infinite three-dimensional networks of the complex ion [Cu(NC–CH2–CH2–CH2–CH2–CN)2]nn+ and nitrate ions. Each copper atom is tetrahedrally surrounded by four nitrogen atoms in this diamond-like open-framework structure.
Following the same procedure, many similar structures have been designed [4–7]. However, the frameworks were very fragile. They collapsed in the absence of guest molecules such as solvents. This hindered their application in catalysis, separation, gas storage, or molecular recognition.
Yaghi’s team found that weak M–N(pyridine, nitrile)-type links made them chemically unstable. So instead of a neutral ligand, a ligand that is –1 charged and a charged metal ion were introduced. “That bond is a lot stronger than a metal nitrile or metal bipyridine bond. It is almost as strong as a carbon–carbon bond”, Omar Yaghi explained.
In 1999, Yaghi and his group finally synthesized a metal-organic framework which remains crystalline and stable when fully desolvated and when heated. The surface area and pore volume of this three-dimensional framework broke all records, it was higher than those of most porous crystalline zeolites [9]. That stimulated the interest in these previously “useless” molecules.
As this new class of porous materials was synthesized from molecular building blocks, it became possible to systematically design properties such as pore size, surface area, the selective uptake of small molecules, and optical or magnetic responses to the inclusion of guests [10].
An amazing example Omar Yaghi gave for the application of MOFs is to capture water from dry air [11]. Using the metal−organic framework (MOF)-801, [Zr6O4(OH)4(fumarate)6], water can be taken up from the atmosphere, be concentrated, and delivered as water drops. The device operates in an exceptionally arid climate (10–40 % relative humidity) and at sub-zero dew points (Tempe, Arizona, USA) with a thermal efficiency (solar input to water conversion) of ~14 %. Yaghi predicted that this device can deliver over 0.25 L of water per kg of MOF for a single daily cycle.
Another application Omar Yaghi spoke about is using MOFs in tanks for methane-powered vehicles [12]. Three times the amount of natural gas can be stored in a tank containing a MOF compared to a tank that does not under the same temperature and pressure. However, with the dropping oil price, the cost for natural gas compared to liquid fuel is not significant enough to make methane-powered vehicles very appealing to consumers. This work is on hold.
Figure 2. During their talks (left to right): Professor Christopher Dobson, University of Cambridge, UK, talked about the amyloid state of proteins and its significance in biology and medicine in particular with respect to Alzheimer’s disease, Professor Frances Arnold, California Institute of Technology (CalTech), Pasadena, USA, talked about designing enzymes for catalysis, and Professor Omar Yaghi talked about reticular chemistry. Pictures: © Royal Society of Chemistry/ MPP.
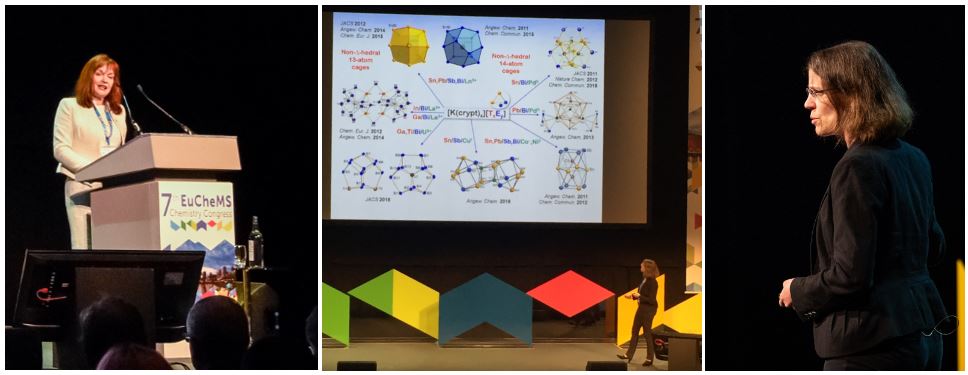
Figure 3. During their talks (left to right): Professor Carol Robinson, University of Oxford, UK, and President of the RSC, talked about using mass spectrometry for folded proteins.
Professor Stefanie Dehnen, Philipps-University of Marburg, Germany, talked about multinary clusters – between exotic structures and potential use. Pictures: © Royal Society of Chemistry/ MPP.
|
Turning Numbers Into Chemical Insight
In his talk, he illustrated his approach using the photochemistry of DNA and conjugated polymers as examples. In DNA, the mechanisms by which UV light is absorbed by interacting nucleobases include localized and collective excitations that give rise to charge-transfer states. As DNA is a large molecule and structurally flexible, it is necessary to compute several thousand excited states using different nuclear configurations to get statistically meaningful results. Analyzing these by hand is tedious at best and can give rise to ambiguous and erroneous conclusions at worst. To circumvent this, it is good to think of each excitation as a connection between an electron and a hole and to analyze the relationship between these instead. In this way, a two-dimensional map of the various states can be constructed. Such a transition density matrix (TDM) can then give a clear picture of the distribution of the various excitations as well as their origin. The TDM for polyadenine shows that nearest-neighbour (DL = 2) interactions are most important, with localized states (DL = 1) and interactions between three bases (DL = 3) also important, but interactions over longer distances are negligible. For a conjugated polymer, such as oligothiophene, a similar approach yields an electron-hole correlation plot, which gives a clear visualization of the excitonic structure. Although the overall electron and hole densities are delocalized for the first excited state, when conditional densities are observed, the electron densities follow the holes. For the second excited state, these correlations are even stronger. However, for the third excited state, the correlation between electron and hole densities is negative. All in all, the electron-hole correlation plot gives us a much clearer and chemically intuitive picture than trying to make sense of delocalized π orbitals. After listening to this talk, I am convinced that a more systematic and statistically inspired approach to analyzing the data generated by quantum chemical calculations will give us more insight. If you would like to apply these methods to your own research, have a look at TheoDORE, an open-source package for wavefunction analysis that plugs into many of the widely used computational programmes (http://theodore-qc.sourceforge.net) or libwfa, also open-source, with interfaces to Q-Chem (single-reference methods) and MOLCAS (multi-reference methods) at https://github.com/libwfa/libwfa. |
Figure 4. Networking breaks and poster sessions. Pictures: © Royal Society of Chemistry/MPP.
EuChemS Announced First Two Historic Landmarks
Brigitte van Tiggelen, Science History Institute, Belgium, held the Public Lecture of the congress. She pointed out that chemistry is an integral part of the chemical heritage of Europe and explained the general idea behind landmark programs to communicate chemistry to a general public. Chemical societies like the German Chemical Society (GDCh), the RSC, and the American Chemical Society (ACS) have already organized various landmark programs on national levels.
This year a new European program has been initiated to honor people or places who or which have had a major impact on chemistry in Europe on a regional level (Category 1) or on a European level (Category 2). Van Tiggelen announced the first European Landmarks:
- ABEA Industries, the oldest olive oil enterprise in Greece, located in Chania on Crete, was founded by the French Chemist Jules Dey in 1889. It is the first European Landmark on the regional level. ABEA applies chemical principles and vertical integration so that all parts of the olive oil harvest can be used. One could say, sustainable chemistry since centuries at its best.
- A ytterbium mine in Sweden is awarded on the European Level. At Grube Ytterby, Vaxholm on the Island of Resaro, East of Stockholm, nine new elements, mostly rare elements, were discovered in the 19th century by chemists from Europe, particularly. A fitting choice in view of next year’s International Year of the Periodic Table (IYPT2019).
Awards and Special Activities
GDCh’s August Wilhelm von Hofmann Medal
Professor Michael Grätzel, École Polytechnique Fédérale de Lausanne (EPFL), Switzerland, has been awarded the August Wilhelm von Hofmann Medal by the Gesellschaft Deutscher Chemiker (GDCh, German Chemical Society). The Medal is awarded every two years for outstanding achievements in chemistry, in particular by scientists working outside of Germany. Michael Grätzel is honored for his pioneering work on low-cost dye-sensitized solar cells (DSSCs), or Grätzel cells. Professor Joachim Maier, Max Planck Institute for Solid State Research in Stuttgart, Germany, held the laudatio with many personal comments.
Figure 5. Professor Jin-Quan Yu (right in the left picture), Scripps Research Institute, La Jolla, CA, USA, receives the 2017 Pedler Award of the RSC for the development of pioneering methods of C–H activation from Professor Sir John Holman, Past-President of the RSC from University of York, UK.
Professor Michael Grätzel (left in the right picture) receives the August Wilhelm von Hofmann medal from Dr. Matthias Urmann, President of the GDCh from Sanofi-Aventis Deutschland GmbH. Pictures: © Royal Society of Chemistry/ MPP.
European Young Chemists‘ Award (EYCA)
Continuing the strong tradition, the European Young Chemists‘ Award (EYCA) session was very well attended again in Liverpool. During the session, twelve preselected Early Career Researchers under the age of 35 and six Ph.D. researchers presented their work and answered questions from the jury comprising Professor Christophe Coperet, ETH Zurich, Switzerland, Professor Luisa De Cola, University of Strasbourg, France, and Professor Gaetano Guerra, University of Salerno, Italy, and from active audience members.
The winners were honored as part of the closing ceremony by the event organizer and coordinator Professor Bruno Pignataro, University of Palermo, Italy: At the Ph.D. level: Erica Del Grosso, University of Rome, Italy, received a special mention, silver medals went to Clément Larquet, Sorbonne Université, France, and Lichen Liu, Universitat València, Italy, and the gold medal was awarded to Tim Gatzmeier from the Max Planck Institute for Coal Research, Mülheim, Germany, for work on activation of challenging substrates with strong chiral Lewis acids.
At the under 35-year-old level, silver medals were awarded to Simone Fabiano, Linköping University, Sweden, and to Matteo Atzori, University of Florence, Italy, and the gold medal and the main award went to Alessandro Porchetta, University of Rome, Italy, for work on programmable nucleic acid nanoswitches for the rapid single-step detection of antibodies in bodily fluids.
Figure 6: (Left to right) Alice Soldà (Chair of EYCN), Alessandro Porchetta, Matteo Atzori, Simone Fabiano, Tim Gatzmeier, Lichen Liu, Clément Larquet, Erica Del Grosso, Pilar Goya (President of EuChemS), Bruno Pignataro. Picture: © Wiley-VCH/NC.
ChemPubSoc Europe Fellows Dinner
ChemPubSoc Europe initiated its Fellows Program in 2013. ChemPubSoc Europe fellows are recognized as pioneers and strong supporters of the ChemPubSoc Europe journals and the publishing activities of the 16 societies involved. Carrying on this tradition, new ChemPubSoc Europe Fellows were announced and a get-together for Fellows, Editors, Board Members, Young Chemists, and other guests took place on Tuesday evening in Liverpool. A fantastic networking event for the diverse members of the chemistry community. The Young Chemists from around the world and their newest member Prof. Dr. Chem Chicken added the fun factor to the event.
|
They are Jolly Good Fellows–Which Nobody Can Deny All agreed that it’s good chemistry which will help to overcome present political challenges in these days of Brexit coming closer and closer with its many unknowns. Initial plans for the next EuChemS Chemistry Congress were revealed and discussed by the representatives of the Chemical Society of Portugal and the president of the Scientific Committee, Luisa De Cola. |
.png)
Figure 7. Snapshots of the ChemPubSoc Europe Reception. Pictures © Wiley-VCH/NC, HR, EW, VK.
Figure 8. The Congress Dinner took place at the spectacular Liverpool Cathedral. Pictures: © Wiley-VCH/VK.
The 8th EuChemS Chemistry Congress (8ECC) will be held in Lisbon, Portugal, from August 30 to September 3, 2020. The meeting will be organized by the Portuguese Chemical Society (SPQ) with the support of the Portuguese Electrochemical Society (SPE). The unifying theme will be “Chemistry the Central Science”.
- 7th EuCheMS Chemistry Congress
Liverpool, UK, August 26-30, 2018 - ChemPubSoc Europe’s activities at EuChemS 2018
- August Wilhelm von Hofmann Medal for Michael Grätzel,
ChemViews Mag. 2018.
Solar cell pioneer honored by Gesellschaft Deutscher Chemiker (GDCh, German Chemical Society) - First European Chemistry Gold Medal,
ChemViews Mag. 2018.
Ben Feringa honored for exceptional achievements in the field of chemistry
References
[1] Reiner Salzer, Philip Taylor, Nineta H. Majcen, Francesco De Angelis, Sophie Wilmet, Evangelia Varella. Ioannis Kozaris, Guest Editorial: The Professional Status of European Chemists and Chemical Engineers, Chem. Eur. J. 2015. https://doi.org/10.1002/chem.201501364
[2] Gilbert N. Lewis, THE ATOM AND THE MOLECULE, J. Am. Chem. Soc. 1916, 38(4), 762. https://doi.org/10.1021/ja02261a002
[3] Y. Kinoshita, I. Matsubara, T. Higuchi, Y. Saito, The crystal structure of bis(adiponitrilo)copper(I) nitrate, Bull. Chem. Soc. Jpn. 1959, 32, 1221–1226. https://doi.org/10.1246/bcsj.32.1221
[4] Alexander Aumüller, Peter Erk, Gerhard Klebe, Siegfried Hünig, Jost Ulrich von Schütz, Hans‐Peter Werner, A Radical Anion Salt of 2,5‐Dimethyl‐N,N′‐dicyanoquinonediimine with Extremely High Electrical Conductivity, Angew. Chem. Int. Ed. 1986. https://doi.org/10.1002/anie.198607401
[5] Bernard F. Hoskins, Richard Robson, Infinite polymeric frameworks consisting of three dimensionally linked rod-like segments, J. Am. Chem. Soc. 1989. https://doi.org/10.1021/ja00197a079
[6] Omar M. Yaghi, D. A. Richardson, G. Li, C. E. Davis, T. L. Groy, Open-Framework Solids With Diamond-Like Structures Prepared From Clusters And Metal-Organic Building Blocks, Mat. Res. Soc. Symp. Proc. 1995, 371.
[7] Omar M. Yaghi, Markus J. Kalmutzki, Christian S. Diercks, Introduction to Reticular Chemistry, Metal-Organic Frameworks and Covalent Organic Frameworks, 1. Edition, Wiley-VCH, Weinheim, Germany, 2019. ISBN: 978-3-527-34502-1
[9] Hailian Li, Mohamed Eddaoudi, M. O’Keeffe, O. M. Yaghi, Design and synthesis of an exceptionally stable and highly porous metal-organic framework, Nature 1999, 402, 276–279. https://doi.org/10.1038/46248
[10] M. Eddaoudi, J. Kim, N. Rosi, D. Vodak, J. Wachter, M. O’Keeffe, O. M. Yaghi, Systematic design of pore size and functionality in isoreticular MOFs and their application in methane storage, Science 2002, 295(5554), 469–472. https://doi.org/10.1126/science.1067208
[11] Hyunho Kim, Sameer R. Rao, Eugene A. Kapustin, Lin Zhao, Sungwoo Yang, Omar M. Yaghi, Evelyn N. Wang, Adsorption-based atmospheric water harvesting device for arid climates, Nature Communic. 2018. https://doi.org/10.1038/s41467-018-03162-7
[12] Mohamed Eddaoudi, Jaheon Kim, Nathaniel Rosi, David Vodak, Joseph Wachter, Michael O’Keeffe, Omar M. Yaghi, Systematic Design of Pore Size and Functionality in Isoreticular MOFs and Their Application in Methane Storage, Science 2002, 295(5554), 469–472. https://doi.org/10.1126/science.1067208




 At the recent EuChemS meeting in Liverpool, I had the pleasure of attending a talk by Dr. Felix Plasser, Lecturer in Chemistry at Loughborough University, entitled “Analysis of Excited-State Computations: Turning Numbers into Chemical Insight”. Methods in computational chemistry have come a long way in the last couple of decades, so much so that almost any 3–5-letter combination can be ascribed to a force field, an ab initio method or a density functional, to paraphrase Dr. Plasser. Coupling this to excellent environmental models and a massive increase in algorithmic efficiency, it means that we have an unprecedented ability to calculate the properties of ever-larger molecules and systems to a level of accuracy that was unthinkable even just ten years ago. However, the sad fact is that, after doing one of these state-of-the-art calculations, we often end up making only a qualitative assessment of the HOMO and LUMO and perhaps checking a few structural parameters. This is, indeed, a sorry state of affairs and the focus of Dr. Plasser’s work is to change this.
At the recent EuChemS meeting in Liverpool, I had the pleasure of attending a talk by Dr. Felix Plasser, Lecturer in Chemistry at Loughborough University, entitled “Analysis of Excited-State Computations: Turning Numbers into Chemical Insight”. Methods in computational chemistry have come a long way in the last couple of decades, so much so that almost any 3–5-letter combination can be ascribed to a force field, an ab initio method or a density functional, to paraphrase Dr. Plasser. Coupling this to excellent environmental models and a massive increase in algorithmic efficiency, it means that we have an unprecedented ability to calculate the properties of ever-larger molecules and systems to a level of accuracy that was unthinkable even just ten years ago. However, the sad fact is that, after doing one of these state-of-the-art calculations, we often end up making only a qualitative assessment of the HOMO and LUMO and perhaps checking a few structural parameters. This is, indeed, a sorry state of affairs and the focus of Dr. Plasser’s work is to change this.