α,ω-Dicarboxylic acids (DCAs, pictured above) are widely used building blocks in the manufacture of, e.g., polymers, plasticizers, lubricants, surfactants, and drugs. DCAs can be produced by disruptive oxidation of ricinoleic acid, a common fatty acid found in hydrolyzed castor oil. However, this route uses strong acids, such as H2SO4 and HNO3, or toxic oxidants. It also only works efficiently under high temperatures and pressures, which raises environmental and safety concerns.
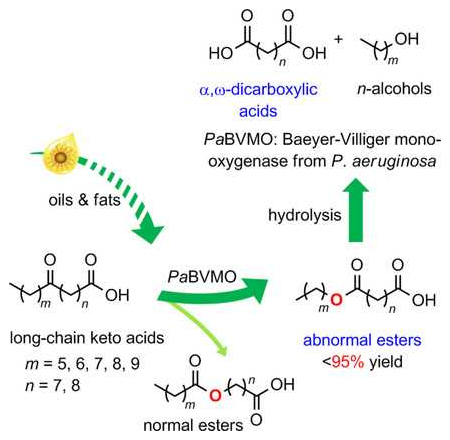
Yunpeng Bai, Jian-He Xu, and colleagues, East China University of Science and Technology, Shanghai, have discovered a Baeyer-Villiger monooxygenase (BVMO) from Pseudomonas aeruginosa, named PaBVMO, which has a highly abnormal regioselectivity towards long-chain aliphatic keto acids.
BVMOs usually add an oxygen atom adjacent to a carbonyl group in the higher-substituted site of asymmetric ketones (pictured as “normal esters” below). The newly discovered BVMO oxygenates asymmetric keto acids at the lower-substituted end and, therefore, yields products that can be hydrolyzed to form α,ω-dicarboxylic acids directly without additional oxidation (pictured as “abnormal esters” below). This reduces reaction steps and lowers the environmental load.
Based on this new enzyme, the team has developed a chemo-enzymatic route to synthesize sebacic acid, a typical raw material for polyamide, from plant-oil derived keto fatty acids. The reaction has an unusually high regioselectivity of 94 %. According to the researchers, the catalytic performance of BVMO could be further improved with the help of protein engineering. The enzyme could ultimately be used for the industrial biotransformation of renewable feedstocks such as oils and fats to useful chemical products under environmentally friendly conditions.
- Direct Access to Medium-Chain α,ω-Dicarboxylic Acids by Using a Baeyer-Villiger Monooxygenase of Abnormal Regioselectivity,
Jia-Mei Yu, Yuan-Yang Liu, Yu-Cong Zheng, Hao Li, Xiao-Yan Zhang, Gao-Wei Zheng, Chun-Xiu Li, Yun-Peng Bai, Jian-He Xu,
ChemBioChem 2018.
https://doi.org/10.1002/cbic.201800318