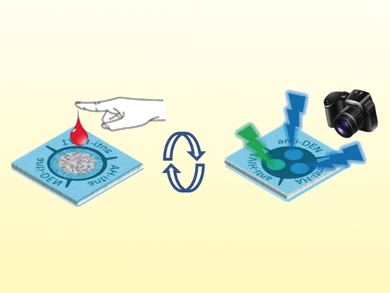
A new paper-based microfluidic device works as a “sample in, signal out” device for the analysis of a single drop of blood. The device was developed by Maarten Merkx, Eindhoven University of Technology, The Netherlands, Daniel Citterio, Keio University, Yokohama, Japan, and colleagues.
The team explains how since George Whitesides and his team turned over a new leaf with paper-based microfluidics more than a decade ago, research has come a long way. These devices replace sophisticated and often expensive standard microfluidics systems made from glass or plastic micro-capillaries with low-cost, lightweight, and disposable systems that use paper instead.
Using Bioluminescence
The researchers have demonstrated how a paper-based device can be built to use bioluminescence resonance energy transfer (BRET) switches for analyte recognition and colorimetric signal generation. “Our devices make use of BRET-based antibody sensing proteins, integrated into vertically assembled layers of functionalized paper,” the team explains.
The first layer is a plasma separation membrane, where cellular components are separated from the whole blood. The second layer contains furimazine as the substrate. The liquid flow then carries all of the components into the third, sensing layer containing the BRET-switching protein LUMABS (luminescent antibody sensor).
“The device design enables sample-volume-independent and fully reagent-free operation, including on-device blood plasma separation.” As a proof of principle, the team showed how it is possible to carry out simultaneous colorimetric detection and quantification of multiple antibodies as model targets: anti-HIV1, anti-HA, and anti-DEN1. This was done with a single drop of whole blood.
Laminated Environment
Because the fully laminated devices provide a closed environment wherein the paper layers absorb a fixed amount of liquid, the sample volume is irrelevant above 20–30 µL. Since all assay components are already integrated into the functionalized paper layers, there is no need for any additional reagents. Indeed, it is an essentially hands-off device once the sample drop has been applied.
A photograph is simply taken 20 minutes after sample introduction to record the bioluminescence. There is no need for any sophisticated pipetting, no liquid handling, nor any analytical equipment other than a camera. As with Whitesides’ original approach in pioneering paper-based microfluidics, the team points out how the simplicity of this type of device would be ideally suited for user-friendly point-of-care-testing in places where resources are sparse.
The team points out that while alternative methods such as ELISA and LUMINEX antibody assays have many uses in analysis and diagnostics, these methods require multiple liquid handling steps. Thus, they have not proven amenable to a transfer on to paper-based systems that could be useful in the developing world and in other cash-strapped healthcare environments. The developed approach also circumvents the sensitivity limitations of lateral-flow immunochromatographic assays.
Functional First
“To the best of our knowledge, this is the first demonstration of a fully integrated paper-based analytical system relying on ratiometric bioluminescence detection in a single drop of whole blood in a highly user-friendly ‘just add the sample’ manner,” the team writes. They suggest that their development offers a way forward in taking experimental laboratory-based techniques in this field into the commercial environment and into healthcare practice in the wider world.
- Paper-Based Antibody Detection Devices Using Bioluminescent BRET-Switching Sensor Proteins,
Keisuke Tenda, Benice van Gerven, Remco Arts, Yuki Hiruta, Maarten Merkx, Daniel Citterio,
Angew. Chem. Int. Ed. 2018.
https://doi.org/10.1002/anie.201808070