Transition-metal-mediated visible-light photocatalysis has opened up new possibilities in synthetic chemistry. In general, the photocatalysts used for this are based on rare and expensive late transition metals, such as ruthenium (Ru) or iridium (Ir). The use of the more abundant lanthanides, in contrast, is underexplored.
Frank Glorius, Westfälische Wilhelms-Universität Münster, Germany, and colleagues have used gadolinium photocatalysis in synthetic photochemistry. The team found that a simple and commercially available lanthanide salt, Gd(OTf)3 (OTf = triflate), can catalyze a photoinduced [2+2] cycloaddition/ring-expansion sequence with indoles. This reaction provides easy access to cyclopenta[b]indoles and indolines. These products are privileged scaffolds found in many bioactive natural products and drug candidates.
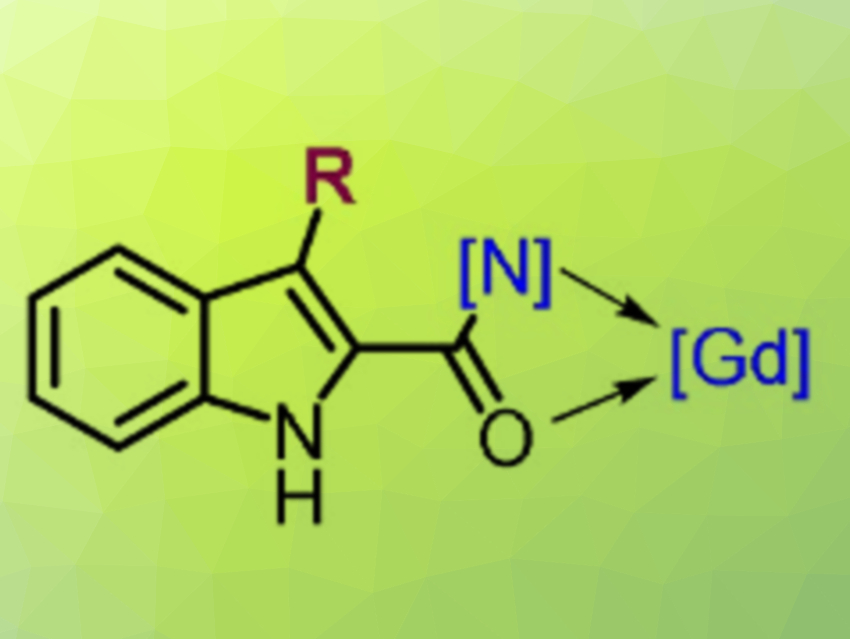
Mechanistic investigations revealed that the Gd(OTf)3 salt plays a key role in forming a photoactive Gd(III) intermediate (pictured). According to the researchers, this implementation of gadolinium photocatalysis could inspire further advancements in photochemistry.
- Gadolinium Photocatalysis: Dearomative [2+2] Cycloaddition/Ring-Expansion Sequence with Indoles,
Jiajia Ma, Felix Schäfers, Constantin Daniliuc, Klaus Bergander, Cristian A. Strassert, Frank Glorius,
Angew. Chem. Int. Ed. 2020.
https://doi.org/10.1002/anie.202001200