Bacteria can form biofilms, which makes them more difficult to treat. The films are composed of bacteria cells embedded in networks of polysaccharides, nucleic acids, and proteins. These networks are important to the mechanical integrity of biofilms and to their increased resistance to antibiotic activity.
Liraz Chai, The Hebrew University of Jerusalem, Israel, and colleagues have analyzed the structure of a bacterial extracellular protein, TapA, under different solution conditions. TapA is secreted by the soil bacterium Bacillus subtilis and plays a role in the formation of biofilms. TapA mediates the assembly of another protein, the functional amyloid protein TasA, into fibrous networks around the cells. It also anchors these fibers to the surface of cells. The team used circular dichroism (CD) and NMR spectroscopy to analyze the structure of TapA.
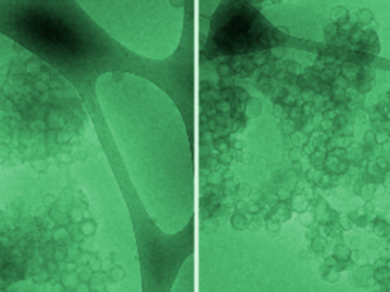
They found that TapA is composed of two domains: a disordered C-terminal domain and a more structured N-terminal domain. Surprisingly, TapA and its two domains were not melted with increasing temperature but rather, they gained some α-helical structure. This process is probably entropically driven and can be explained by the release of water molecules. The two domains of TapA weakly interact with each other in solution. However, they can cooperatively interact, e.g., with lipid vesicles (pictured), which were chosen as a model binding partner for the protein.
These findings suggest that the two-part composition of TapA is important for its interactions in extracellular networks in Bacilli biofilms. Being flexible to external conditions and being composed of two domains may allow TapA to bind to different targets in the extracellular matrix and serve as a cross-linker between TasA and the cell membranes. These results could lead to the design of anti-biofilm drugs that target these networks.
- The bacterial extracellular matrix protein TapA is a two-domain partially disordered protein,
Liraz Chai, Razan Abbasi, Reem Mousa, Noa Dekel, Hadar Amartely, Tsafi Danieli, Mario Lebendiker, Yael Levi-Kalisman, Deborah E. Shalev, Norman Metanis,
ChemBioChem 2018.
https://doi.org/10.1002/cbic.201800634