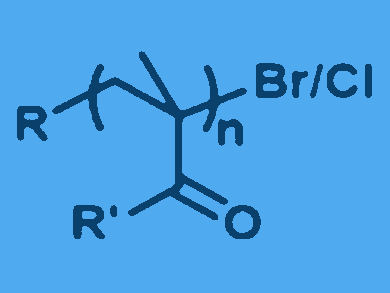
Atom transfer radical polymerization (ATRP) allows the polymerization of a broad selection of monomers with control over the molecular weight, dispersity, and architecture. However, oxygen can poison the activators for ATRP. It can also quench chain growth by forming very stable peroxy radicals with the propagating chains. Therefore, ATRP is typically performed under air-free conditions or with an excess of reducing agents.
Krzysztof Matyjaszewski and colleagues, Carnegie Mellon University, Pittsburgh, USA, have used an enzymatic cascade to remove oxygen and to generate a steady flow of radicals, which are needed for the formation of activators. In this way, the team was able to trigger ATRP in air. After the complete removal of oxygen, the polymerization slowed down or stopped, which contrasts dramatically with conventional polymerizations.
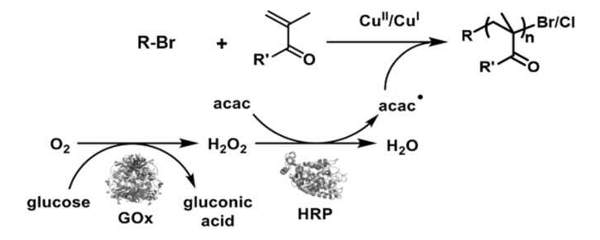
The biocatalytic system (pictured) contains glucose oxidase (GOx), horseradish peroxidase (HRP), and a copper complex. In the presence of glucose and acetylacetonate (acac) as sacrificial substrates, a very stable copper tris(2-pyridylmethyl)amine complex is activated that controls radical polymerization. The team applied the procedure to the synthesis of protein-b-polymer and DNA-b-polymer bioconjugates, and to well-defined polymers and coatings under atmospheric conditions and at room temperature.
- Biocatalytic “Oxygen-Fueled” Atom Transfer Radical Polymerization,
Alan E. Enciso, Liye Fu, Sushil Lathwal, Mateusz Olszewski, Zhenhua Wang, Subha R. Das, Alan J. Russell, Krzysztof Matyjaszewski,
Angew. Chem. Int. Ed. 2018.
https://doi.org/10.1002/anie.201809018