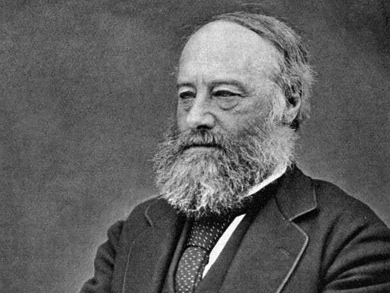James Prescott Joule was born on December 24, 1818, in Salford, UK. He was homeschooled; one of his teachers was the famous chemist John Dalton. Joule was keenly interested in science and liked to experiment, particularly with electricity. He received no formal university education in science.
Joule’s family owned a brewery, and he set up a laboratory in the brewery’s cellar. He considered replacing the steam motors used in the business with electric ones, and in 1838, he began to investigate which type of engine might be more efficient. This research into electric engines led to his discovery of Joule’s first law in 1840 (published in 1841 [1]), which states that the heat (Q) produced by a conductor is proportional to the square of the current intensity (I) that passes through it, multiplied by the resistance (R):
![]()
Joule’s experiments with electricity also led him to his early work on thermodynamics, the branch of physics that deals with the connection between heat and other forms of energy. At the time, it was thought that heat could not be created or destroyed. However, heat is produced and not just transferred when a current is passed through a conductor. Joule figured that different types of energy, including heat, can be converted into one another [2]. In 1842, German physician, chemist, and physicist Robert Mayer had found independently that the ratio between the heat produced and the mechanical work required to produce it is constant [3].
Between 1843 and 1847, Joule undertook a variety of experiments to find the value of this constant (today known to be 4.1855 J/cal at 15 °C) [4]. He determined the mechanical equivalent of heat, e.g., by using an electric motor [2], by compressing a gas to heat it [5], and by using a paddle wheel to heat water [6]. All these different types of experiments led to comparable values for the mechanical equivalent of heat. His results were met with skepticism at first due to his lack of formal training, but the mounting experimental evidence gradually convinced his contemporaries [7].
Joule collaborated—mostly by mail—with William Thomson, later known as Lord Kelvin. In 1852, the team showed that an expanding gas cools down, a phenomenon named Joule–Thomson effect in their honor [8]. For an ideal gas without any interactions between the particles, this effect is zero. This is in accordance with Joule’s second law, which states that the specific internal energy of an ideal gas depends only on its temperature, not on volume or pressure [5].
In part due to Thomson’s support, Joule’s work overcame the initial resistance by his peers. Despite never receiving a university education, Joule was named a Fellow of the Royal Society in 1850 and received the Royal Medal in 1852 “for his paper on the mechanical equivalent of heat”, the Copley Medal in 1870 “for his experimental researches on the dynamical theory of heat”, and the Albert Medal in 1880 “for having established, after most laborious research, the true relation between heat, electricity and mechanical work, thus affording to the engineer a sure guide in the application of science to industrial pursuits”.
Joule received three honorary degrees (from Trinity College Dublin, today’s Ireland, the University of Oxford, UK, and the University of Edinburgh, UK), as well as a state pension for his services to science. Joule died on October 11, 1889, in Sale, UK. The SI unit of energy is named joule (J) in his honor.
James Prescott Joule is the answer to Guess the Chemist (84).
References
- [1] On the heat evolved by metallic conductors of electricity, and in the cells of a battery during electrolysis,
James Prescott Joule,
Philos. Mag. 1841, 19, 260–277.
https://doi.org/10.1080/14786444108650416 - [2] On the calorific effects of magneto-electricity, and on the mechanical value of heat,
James Prescott Joule,
Philos. Mag. 1843, 23, 263–276.
https://doi.org/10.1080/14786444308644730 - [3] Bemerkungen über die Kräfte der unbelebten Natur (in German),
Robert Julius Mayer,
Ann. Chem. Pharm. 1842, 233–240.
https://doi.org/10.1002/jlac.18420420212 - [4] IUPAC Gold Book, Calorie, goldbook.iupac.org. (accessed November 2018)
- [5] On the changes of temperature produced by the rarefaction and condensation of air,
James Prescott Joule,
Philos. Mag. 1845, 26, 369–383.
https://doi.org/10.1080/14786444508645153 - [6] On the mechanical equivalent of heat, as determined by the heat evolved by the friction of fluids,
James Prescott Joule,
Philos. Mag. 1847, 31, 173–176.
https://doi.org/10.1080/14786444508645153 - [7] On the mechanical equivalent of heat,
James Prescott Joule,
Phil. Trans. R. Soc. Lond. 1850, 140, 61–82.
https://doi.org/10.1098/rstl.1850.0004 - [8] On the thermal effects experienced by air in rushing through small apertures,
James Prescott Joule, William Thomson,
Philos. Mag. 1852, 4, 481–492.
https://doi.org/10.1080/14786445208647169
Sources
- James Joule: A Biography,
Donald S. L. Cardwell,
Manchester University Press, 1989.
ISBN: 978-0719034794 - James Joule – Brewer and Man of Science,
Alan Gall,
Brew. Hist. 2004, 115, 2–6.
Selected Publications
- The Scientific Papers of James Prescott Joule,
James Prescott Joule,
Physical Society, London 1884.