Water electrolysis is a green and efficient approach to producing hydrogen as a fuel. However, the commercialization of this technology requires high-performance and affordable electrocatalysts for the hydrogen evolution reaction (HER). Cobalt phosphide (CoP) is a promising catalytic material for the HER because of its excellent electrical conductivity and good corrosion resistance.
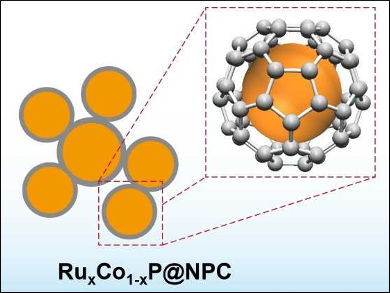
Wei Zhou, Nanjing Tech University, China, Zongping Shao, Nanjing Tech University and Curtin University, Perth, Australia, and colleagues have prepared a nanostructured hybrid of Ru-doped CoP, encapsulated in an N,P-doped ultrathin carbon layer (RuxCo1–xP@NPC, pictured) via a simple and easily scalable procedure. The team used cobalt(II) nitrate, ruthenium(III) chloride, NH4H2PO4, and melamine as reagents. These precursors were annealed at a temperature of 900 °C to prepare the catalyst.
Ru-doped CoP@NPC particles have enhanced activities compared to the undoped counterparts. Ru0.25Co0.75P@NPC showed the best HER activity with a low overpotential of only 73 mV at a current density of 10 mA cm–2 in acidic media. This performance is close to the benchmark catalyst Pt/C.
The team attributes the improved activity to several effects: a) The introduction of Ru into the CoP structure modulates the electronic structure in a favorable way, b) the carbon layer derived from melamine serves as a conductive carrier and also prevents the aggregation of the nanoparticles, c) the strong coupling between the Ru0.25Co0.75P phase and the N,P-doped carbon shell could generate a synergistic effect which favors HER catalytic activity.
- Multifold Nanostructuring and Atomic-Scale Modulation of Cobalt Phosphide to Significantly Boost Hydrogen Production,
Jie Yu, Xinhao Wu, Yijun Zhong, Guangming Yang, Meng Ni, Wei Zhou, Zongping Shao,
Chem. Eur. J. 2018, 24, 13800–13806.
https://doi.org/10.1002/chem.201802667