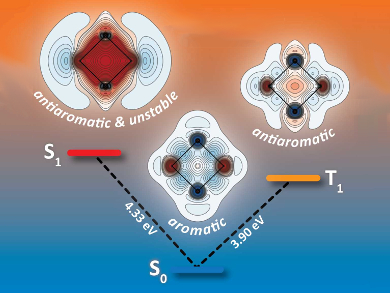
According to the well-known Hückel rule, aromaticity in ground-state conjugated hydrocarbon rings requires 4n+2 π-electrons, while rings with 4n π-electrons are antiaromatic. According to Baird’s rule, these criteria switch in the lowest triplet excited state: Rings with 4n π-electrons become aromatic and rings with 4n+2 π-electrons become antiaromatic. An analogous effect exists for the lowest singlet excited state.
By calculating and analyzing the magnetic shielding surrounding the molecule for each electronic state, Peter B. Karadakov, University of York, UK, and colleagues have shown that S2N2 is the first example of an inorganic ring for which theory predicts aromaticity switching upon electronic excitation. The team used multiconfigurational quantum chemical calculations to find the magnetic shielding values.
The ground state (pictured center) is aromatic, but less so than the archetypal aromatic molecule benzene. The lowest triplet state (pictured right) is antiaromatic, like the singlet ground state of square cyclobutadiene. The first singlet excited state of S2N2 (pictured left) is profoundly antiaromatic, to an extent that removes bonding interactions. These findings provide new directions in the search for molecules with tuneable (anti-)aromaticity for applications in electronics, photovoltaics, and molecular motors.
- Magnetic Shielding, Aromaticity, Antiaromaticity and Bonding in the Low-Lying Electronic States of S2N2,
Peter B. Karadakov, Muntadar A. H. Al-Yassiri, David L. Cooper,
Chem. Eur. J. 2018, 24, 16791–16803.
https://doi.org/10.1002/chem.201804292