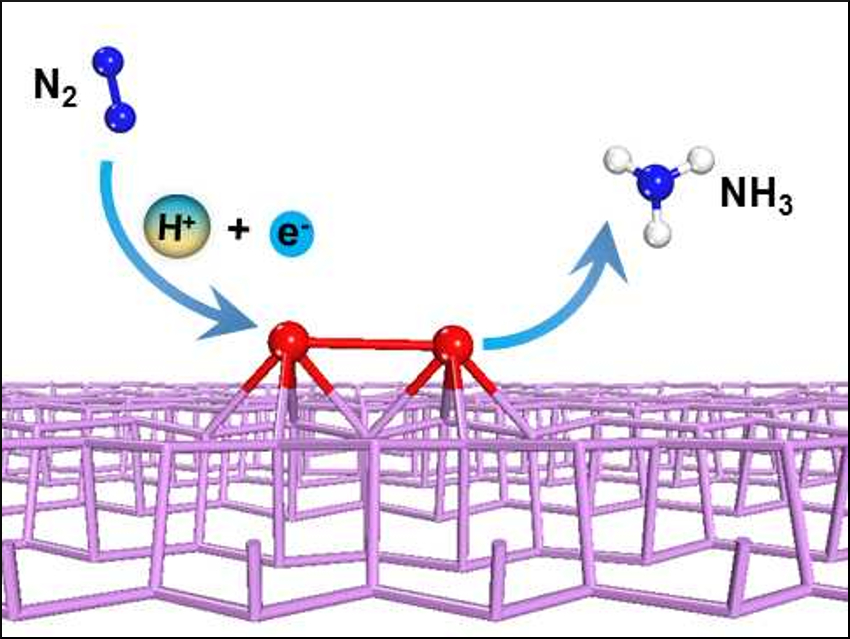
Using electricity to drive nitrogen reduction to ammonia at ambient conditions is a promising alternative to the energy-intensive, high-temperature, and high-pressure Haber–Bosch process. However, finding an electrocatalyst that can effectively activate and convert nitrogen at a low applied voltage is challenging.
Using density functional theory (DFT) calculations, Qing Tang, Chongqing University, China, and De-en Jiang, University of California, Riverside, USA, have predicted that a titanium dimer supported on phosphorene—a 2D single layer of black phosphorus—could be a promising electrocatalyst for nitrogen capture and conversion. The researchers discovered that the strong N–N triple bond is significantly weakened and activated on this material, and that the overpotential needed for ammonia production can be as low as 0.2 V, much lower than that required when using a bulk metal surface or nitrides.
By screening other first-row transition-metal dimers, the researchers found that Sc and Fe dimers also show a high reactivity for nitrogen electroreduction. The study provides a theoretical roadmap for exploring phosphorene-supported few-atom catalysts for nitrogen fixation.
- Phosphorene-Supported Transition-Metal Dimer for Effective N2 Electroreduction,
Qing Tang, De-en Jiang,
ChemPhysChem 2019.
https://doi.org/10.1002/cphc.201900279