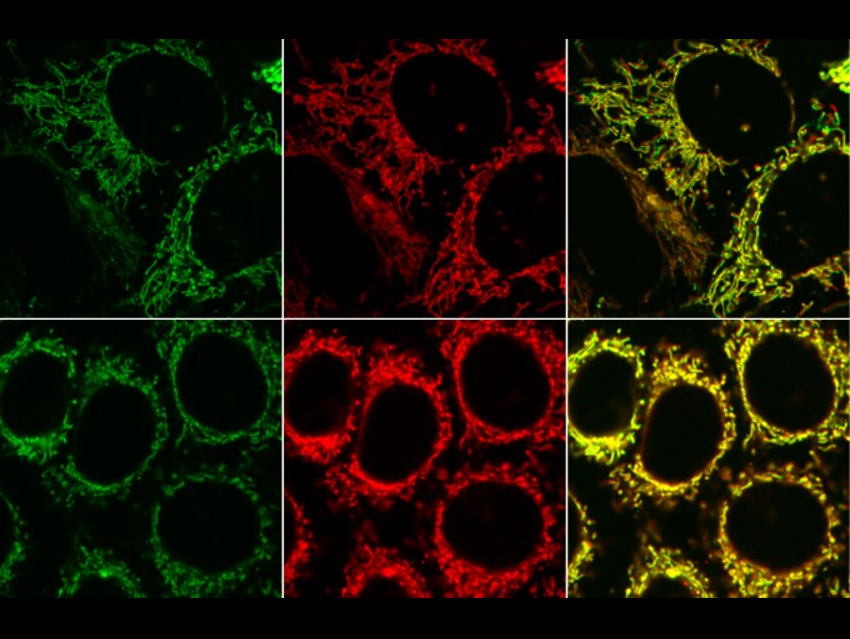
To improve the understanding of cellular biology monitoring individual biomolecules in biological processes is important. One strategy for tracking molecules inside cells is to attach small fluorescent dyes to subsequently detect the compounds of interest by fluorescence microscopy (examples pictured).
However, an undesired background fluorescent signal that stems from the dye unspecifically binding to cellular compartments often complicates such experiments and leads to misinterpretation of the data. Turning on fluorescence only where the labeling is supposed to happen could help with this.
Milan Vrabel and colleagues, Czech Academy of Sciences, Prague, have synthesized fluorescent products from two nonfluorescent starting materials, i.e., that allow fluorescence turn‐on labeling. Bicyclononynes were reacted with 1,2,4,5-tetrazines with electron‐donating substituents. The reaction gives fluorescent 4,5‐dihydropyridazines and can be performed under biological conditions.
The increase in fluorescence intensity can be up to 900‐fold when a symmetric double‐substituted tetrazine is used. This is among the highest values reported for a bioorthogonal reaction to date. The reaction remains fluorogenic even in live cells. According to the researchers, the method can be used to illuminate biomolecules with an improved signal-to-noise ratio.
- Bioorthogonal Fluorescence Turn-On Labeling Based on Bicyclononyne–Tetrazine Cycloaddition Reactions that Form Pyridazine Products,
Sebastian J. Siegl, Juraj Galeta, Rastislav Dzijak, Martin Dračínský, Milan Vrabel,
ChemPlusChem 2019.
https://doi.org/10.1002/cplu.201900176