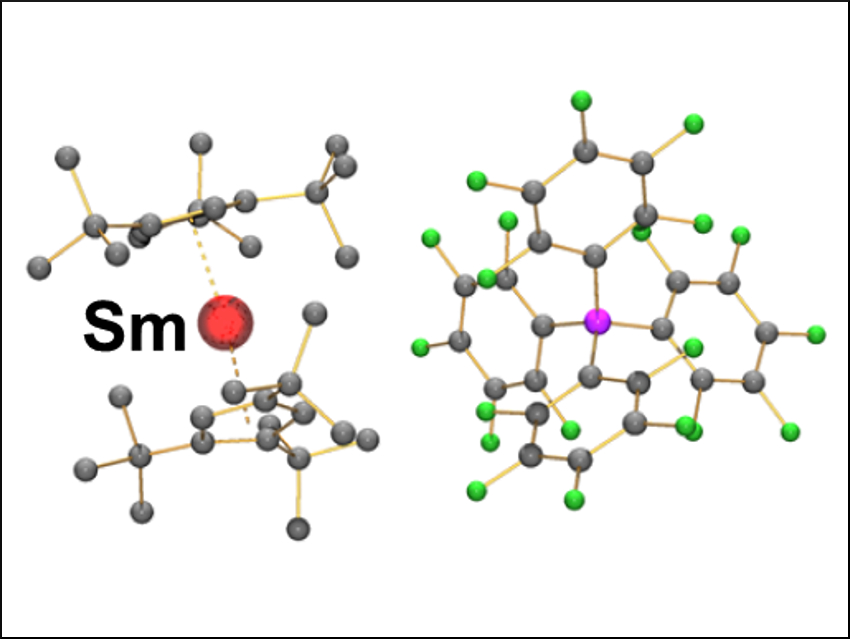
Since ferrocene ([Fe(Cp)2] (Cp = cyclopentadienyl, C5H5)) was first reported in the 1950s [1], structurally similar derivatives (metallocenes) and their cations (metalloceniums) have been widely investigated. Lanthanide metallocenium cations tend to show equatorial interactions with counterions perpendicular to the “sandwich” due to their predominantly ionic bonding and large lanthanide cations. Recently, examples of lanthanide metallocenium cations have been reported that show no equatorial interactions [2]. These compounds have bulky substituents and weakly coordinating anions; in some cases, they have provided remarkable single-molecule magnets.
David Mills, Nicholas Chilton, and colleagues, University of Manchester, UK, have synthesized a series of light lanthanide metallocenium complexes. Samarium is as an example pictured on the right, with its anion on the left. The complexes were synthesized by the reactions of [Ln(Cpttt)2(Cl)] (Ln = La, Ce, Pr, Nd, Sm; Cpttt = C5H2tBu3‐1,2,4) with [H(SiEt3)2][B(C6F5)4] in benzene.
Equatorial interactions are present for lanthanides larger than samarium. This shows that a combination of bulkier cyclopentadienyl ligands and more weakly coordinating anions would be required to achieve isolated lanthanide metallocenium cations for the early lanthanides. The team also studied the physical properties of these complexes, which could be useful for researchers interested in exploiting the magnetic properties of lanthanide metallocenium complexes in the future.
- Light Lanthanide Metallocenium Cations Exhibiting Weak Equatorial Anion Interactions,
Jingjing Liu, Daniel Reta, Jake A. Cleghorn, Yu Xuan Yeoh, Fabrizio Ortu, Conrad A. P. Goodwin, Nicholas F. Chilton, David P. Mills,
Chem. Eur. J. 2019.
https://doi.org/10.1002/chem.201901167
[1] T. J. Kealy, P. L. Pauson, A New Type of Organo-Iron Compound, Nature 1951, 168, 1039–1040. https://doi.org/10.1038/1681039b0
[2] Conrad A. P. Goodwin, Daniel Reta, Fabrizio Ortu, Nicholas F. Chilton, David P. Mills, Synthesis and Electronic Structures of Heavy Lanthanide Metallocenium Cations, J. Am. Chem. Soc. 2017. https://doi.org/10.1021/jacs.7b11535