The nitrogen reduction reaction is important for the production of ammonia. Often, precious metals or transition-metal compounds have been used as catalysts for this reaction. Only a few studies have focused on the used of catalysts based on main-group elements.
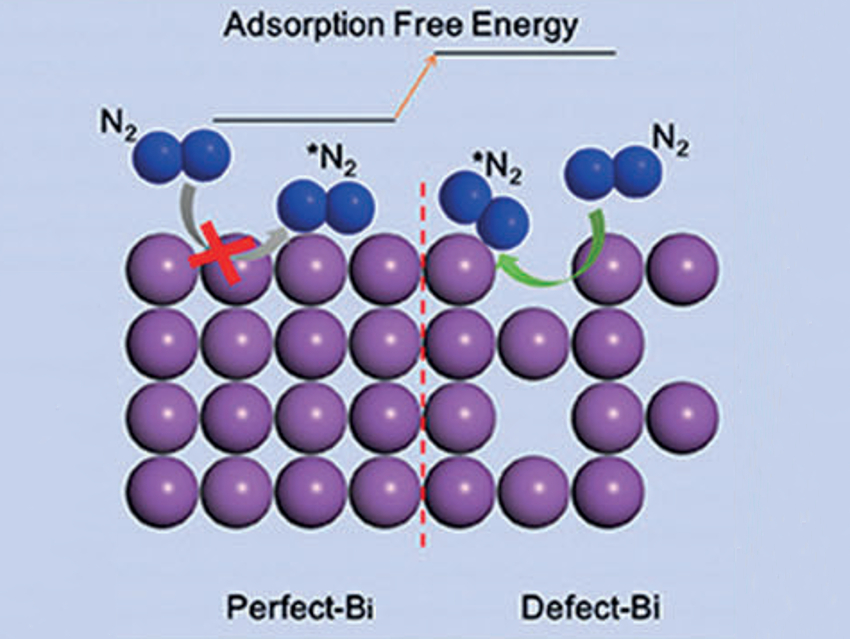
Jun-min Yan, Jilin University, Changchun, China, Jingguang G. Chen, Columbia University, New York, USA, and colleagues thought that bismuth could be useful for the electrochemical nitrogen reduction. The main-group element binds only weakly with hydrogen atoms, which could increase the selectivity and production rate of ammonia. However, the electronic structure of Bi is usually unfavorable for N2 adsorption and activation. This could be changed by introducing defects (pictured right).
The researchers synthesized defect-rich Bi using a low-temperature plasma bombardment technology and characterized the resulting catalysts by positron annihilation and X-ray absorption spectroscopy to verify the existence of defects. The defects play a vital role in enabling the adsorption and activation of dinitrogen (N2) on defect-rich Bi for NH3 production. The catalyst achieved an NH3 production rate of 5.453 μg mgBi−1 h−1 and a Faradaic efficiency of 11.68 %. According to the team, the use of defects to convert a material that is almost inactive for nitrogen reduction into an active catalyst is a promising concept for future research.
- Generating Defect-Rich Bismuth for Enhancing the Rate of Nitrogen Electroreduction to Ammonia,
Yue Wang, Miao-miao Shi, Di Bao, Fan-lu Meng, Qi Zhang, Yi-tong Zhou, Kai-hua Liu, Yan Zhang, Jia-zhi Wang, Zhi-wen Chen, Da-peng Liu, Zheng Jiang, Mi Luo, Lin Gu, Qing-hua Zhang, Xing-zhong Cao, Yao Yao, Min-hua Shao, Yu Zhang, Xin-Bo Zhang, Jingguang G. Chen, Jun-min Yan, Qing Jiang,
Angew. Chem. Int. Ed. 2019.
https://doi.org/10.1002/anie.201903969