Americium is an actinide and the first transplutonium element. Its long‐lived isotope 243Am is most suitable for synthetic chemistry. However, 243Am is both scarce and highly radioactive, which makes it difficult to work with. There are examples of organometallic actinide compounds. However, no transplutonium organometallic compound had been characterized using single-crystal X-ray diffraction so far.
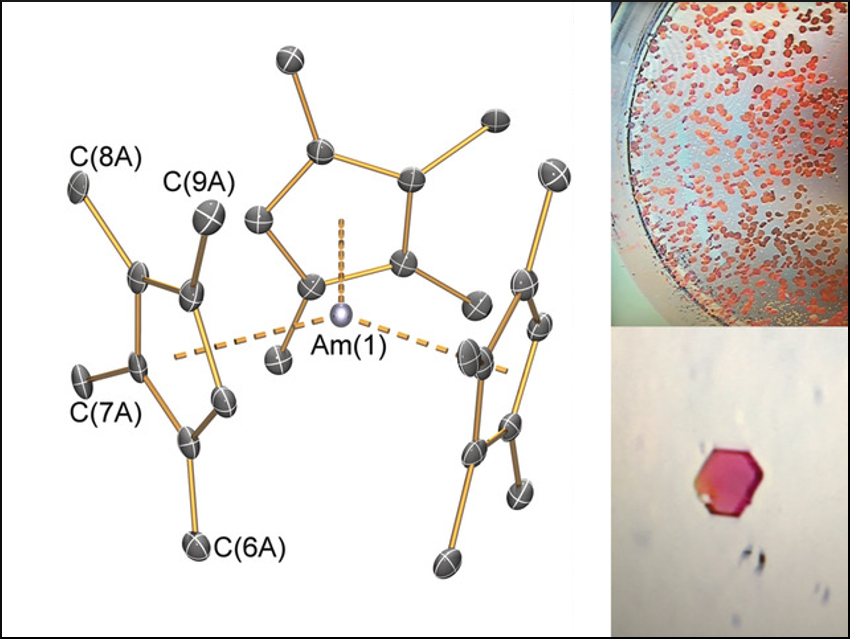
Enrique R. Batista, Andrew J. Gaunt, Stosh A. Kozimor, Ping Yang, Los Alamos National Laboratory, NM, USA, William J. Evans, University of California, Irvine, USA, and colleagues have synthesized the Am3+ organometallic complex [Am(C5Me4H)3] (pictured). Due to the scarcity of 243Am, small-scale optimization was performed on lanthanide and actinide analogues to enable successful work with only 5 mg of Am.
The team started from 243AmO2, which was dissolved in hydrochloric acid and dried under an argon stream to give an AmCl3 residue. Dimethyl ether (DME) and trimethylsilyl chloride (TMSCl) were added to give a putative AmCl3(DME)n reactant. Potassium tetramethylcyclopentadienyl was added to give the desired product in 49 % yield as an orange crystalline solid (pictured). The complex was characterized by single‐crystal X‐ray diffraction and is isostructural with other [M(C5Me4H)3] complexes.
- [Am(C5Me4H)3]: An Organometallic Americium Complex,
Conrad A. P. Goodwin, Jing Su, Thomas E. Albrecht-Schmitt, Anastasia V. Blake, Enrique R. Batista, Scott R. Daly, Stefanie Dehnen, William J. Evans, Andrew J. Gaunt, Stosh A. Kozimor, Niels Lichtenberger, Brian L. Scott, Ping Yang,
Angew. Chem. Int. Ed. 2019.
https://doi.org/10.1002/anie.201905225