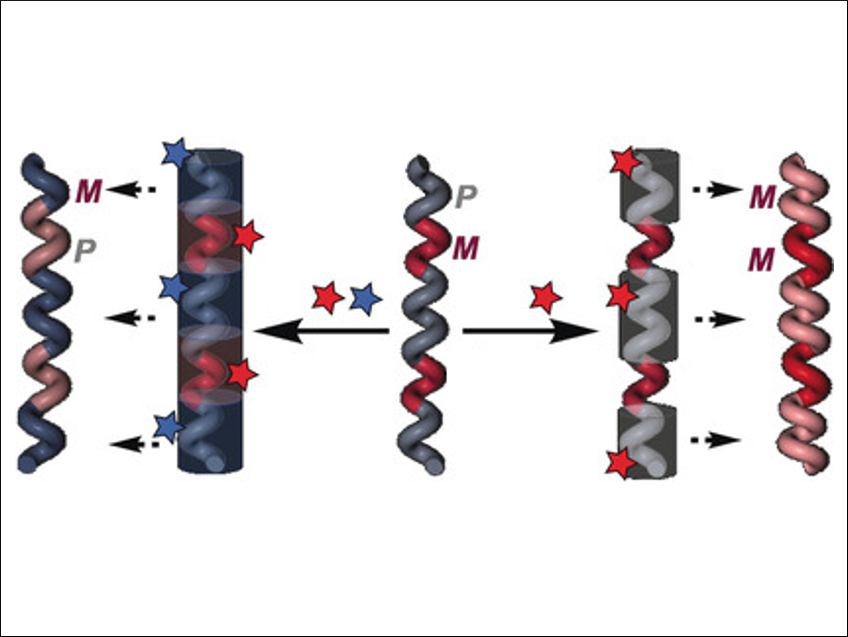
“Chiral conflict” in helical polymers is a phenomenon where two different monomers in a single copolymer induce opposite helical senses. This produces an axially racemic helix. In this field, P denotes a right-handed helix and M denotes a left-handed helix.
Félix Freire and colleagues, Universidade de Santiago de Compostela, Spain, have applied this concept to create stimuli‐responsive materials based on dynamic helical polymers. Using a rhodium norbornadiene chloride catalyst, the team prepared different polymers from the monomers (R)-N-(4-ethynylphenyl)-2-methoxy-2-phenylacetamide (m-(R)-1) and methyl (R)-2-(4-ethynylbenzamido)-2-phenylacetate (m-(R)-2). In the resulting polymers, poly-(R)-1 has an M helix, poly-(R)-2 has a P helix, and in the copolymer poly-[(R)-1x–co-(R)-21-x] (x=0.1–0.9), chiral conflict occurs and a mixture of P and M helices is observed.
The team discovered that when the polymers are exposed to external stimuli, such as the addition of metal perchlorate salts, the helical sense and/or elongation of the polymers can be altered. In a copolymer comprised of P-and M-helix-inducing monomers, a stimulus that selectively interacts with one of the monomers can produce a partial helix inversion. This results in a single-handed helix (pictured right). The preference for M or P helices depends on the metal ion in the perchlorate and on the copolymer composition. When stimuli invert the helicity at both types of monomers, this effect can generate an axially racemic mirror image of the original copolymer (pictured left).
The researchers believe that this chiral-conflict effect can be used to transform axially racemic polymers into new chiral functional materials with applications in sensing, asymmetric synthesis, or chiral recognition.
- Chiral Conflict as a Method to Create Stimuli-Responsive Materials Based on Dynamic Helical Polymers,
Mohammad Alzubi, Sandra Arias, Rafael Rodríguez, Emilio Quiñoá, Ricardo Riguera, Félix Freire,
Angew. Chem. Int. Ed. 2019.
https://doi.org/10.1002/anie.201907069