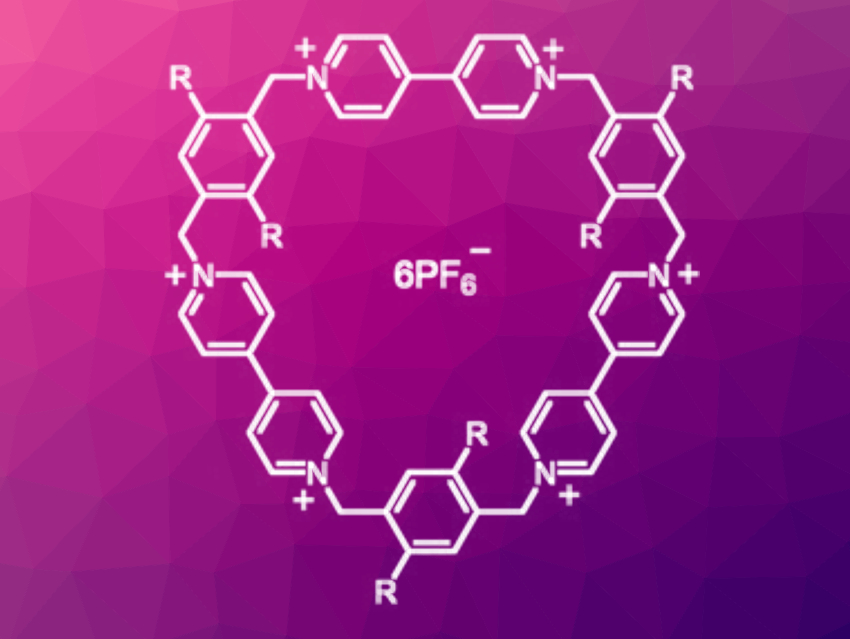
Bipyridinium radical cations can interact with one another through pimerization, i.e., π‐dimerization. Consequently, cationic cyclophanes that contain bipyridinium units can form a variety of supramolecular entities when reduced to their radical forms.
Ommid Anamimoghadam, Northwestern University, Evanston, IL, USA, J. Fraser Stoddart, Northwestern University, Tianjin University, China, and University of New South Wales, Sydney, Australia, and colleagues have synthesized a new class of bipyridinium-containing cyclophanes, cyclotris(paraquat-p-phenylenes) (pictured, R = H, OMe). These bicyclic cyclophanes contain three bipyridinium units, separated by xylylene spacers, in a triangular arrangement. They were synthesized from two fragments by reacting the corresponding bis(bromomethyl-aryl)dipyridinium and (aryl-bismethylene)dipyridinium salts.
X-ray crystal (super)structure analysis shows that the methoxy-substituted derivative forms channel-like architectures. The unsubstituted parent compound crystallizes in two polymorphs in the form of assemblies with intercalated PF6– counterions. In the reduced, radical state of the methoxy-substituted derivative, the team observed stacks between the 1,4-dimethoxyphenylene and bipyridinium radical cations.
These structures show that subtle modifications of redox-active shape-persistent cyclophanes can have significant consequences for their assembly. This work could inspire new directions in the design and study of organic radicals, supramolecular entities, and solid-state materials.
- Cyclotris(paraquat-p-phenylenes),
Ommid Anamimoghadam, James A. Cooper, Minh T. Nguyen, Qing-Hui Guo, Lorenzo Mosca, Indranil Roy, Junling Sun, Charlotte L. Stern, Louis Redfern, Omar K. Farha, J. Fraser Stoddart,
Angew. Chem. Int. Ed. 2019.
https://doi.org/10.1002/anie.201907329