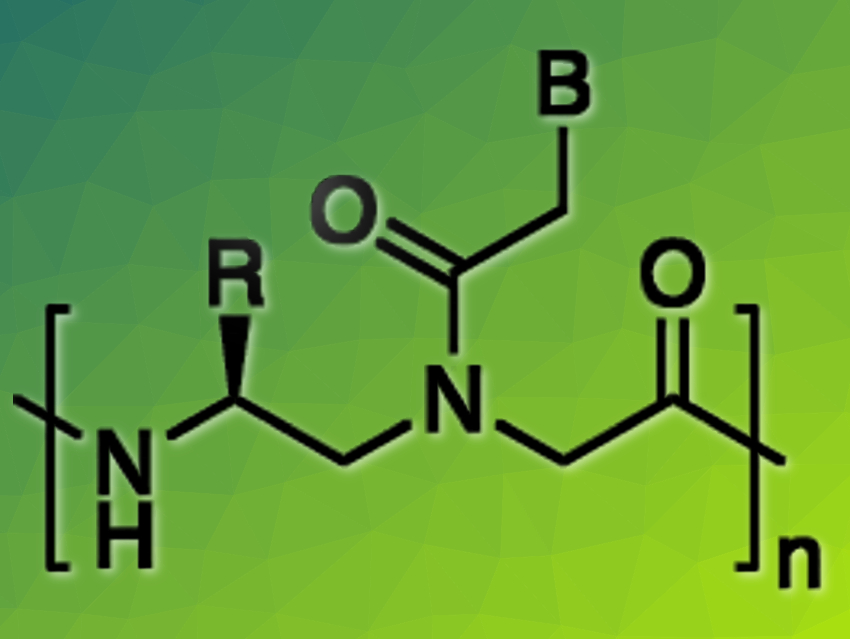
Jennifer M. Heemstra, Emory University, Atlanta, GA, USA, and colleagues have given an oligomeric system the ability to communicate in both of nature’s fundamental languages simultaneously. The new biopolymer, referred to as a peptide nucleic acid or PNA (pictured), combines the functional information contained in the peptide structure with the genetic information given by the nucleobases. It is the first molecule that can readily communicate in both languages and can interact with its environment depending on the language in which it is addressed, say the researchers. Bilingual biopolymers may be helpful as functional adaptors in biomedical or nanotechnology applications.
Nature Talk
Nucleic acids store genetic information and convey it—or “speak”—through base pairing: The nucleobases form hydrogen bonds with their matches in complementary strands. Proteins, in contrast, can only speak with their environment and exert their function when they have obtained the correct three-dimensional structure, which is predefined by the amino acid sequence.
Nature keeps both languages separate. A structurally or functionally active amino acid is never found embedded in a nucleic acid biopolymer, and a genetically encoding nucleobase never mingles with neighboring amino acids within a peptide or protein chain. However, scientists argue that a combination of both codes in one molecule could lead to true bilingualism. A bilingual molecule could be useful in several ways: In biomedicine, for example, it could serve as a hub to adapt to or mediate between incoming signals.
Truly Bilingual
A simple hybrid PNA that includes peptide and nucleic acid sections is not difficult to make—the chemist only has to connect two polymer blocks, one with the base and the other with the protein information. However, the team found that in such a system, both languages run separately and possibly in parallel, but they cannot control each other. Therefore, the team designed a PNA with a more interwoven structure. This protein–nucleobase hybrid will switch between both languages, depending on which information dominates.
As monomers, they synthesized γ-functionalized amino acids that carry both entities in one molecule, a nucleobase (B) and an amino acid side chain (R)—or a fluorescing molecule, which signals the aggregation state of the molecule. If different monomers are joined with peptide bonds, the resulting oligomer contains the base information as well as the functions given by the amino acid side chains that sit between the sequence of bases.
As the first truly bilingual PNA, the researchers created an oligomer of twelve units, with a base sequence matching that of miRNA-21, a biomedical RNA target that is typically upregulated in cancer cells. The team included alanine (methyl group) or lysine (4-aminobutyl group) side chains in the backbone so that the C-terminus of the strand was hydrophobic (two alanines in a row) but the N-terminus was hydrophilic (two lysines).
In aqueous solution, the resulting PNA oligomers self-assembled through their hydrophobic parts to form micelles. This alanine-induced micelle formation indicated that their amino-acid encoded language was dominant because, in a similar way, peptides fold to form three-dimensional structures or aggregates with their neighbors. However, when miRNA-21 or DNA with matching bases was introduced, the strands hybridized, and the micelles broke up. In this case, the molecule switched to the other language.
More Architectures
The researchers admit that their first truly bilingual PNA was still somewhat restricted in its speech: The peptidic language only provided information on solubility, whereas the base sequence was chosen to be pyrimidine-rich—purine bases appeared to thwart PNA synthesis and hybridization.
Despite these challenges, the scientists are convinced that more exploratory work and a wider variety in structural motifs could expand both the structural and genetic language of PNA. Ultimately, they see PNA as a possible mediator or adaptor between medical targets such as proteins and nucleic acids.
- Bilingual Peptide Nucleic Acids: Encoding the Languages of Nucleic Acids and Proteins in a Single Self-Assembling Biopolymer,
Colin S. Swenson, Arventh Velusamy, Hector S. Argueta-Gonzalez, Jennifer M. Heemstra,
J. Am. Chem. Soc. 2019.
https://doi.org/10.1021/jacs.9b09146