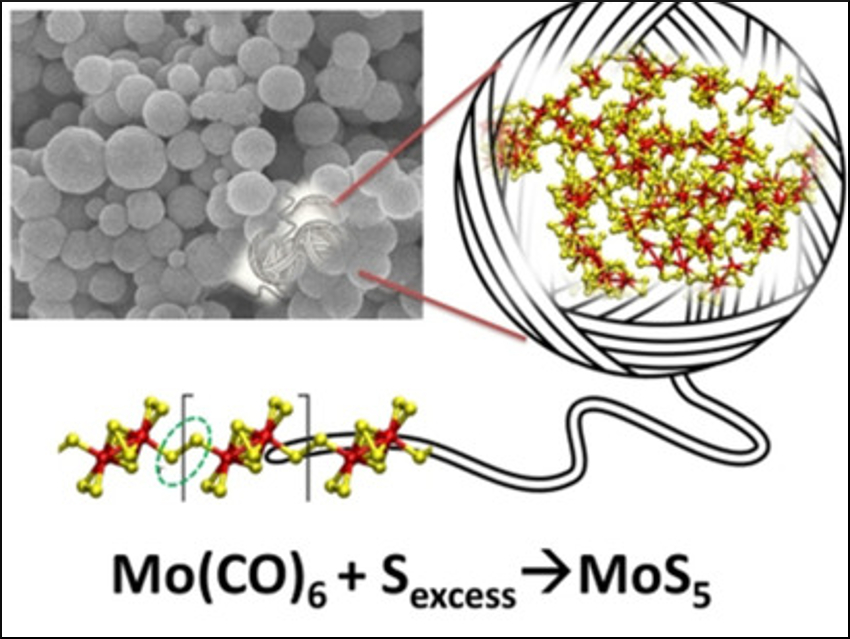
Sulfur‐rich transition-metal polysulfides, such as MoS5, could be useful in catalytic processes, energy storage, etc. due to their abundant disulfide groups (–S–S–). However, many transition-metal polysulfides are amorphous, and their structures, as well as the reasons for their amorphous state, are hard to determine.
Ekaterina D. Grayfer, Nikolaev Institute of Inorganic Chemistry, Novosibirsk, Russia, Sofya B. Artemkina, Nikolaev Institute of Inorganic Chemistry and Novosibirsk State University, Andrey N. Enyashin, Institute of Solid State Chemistry, Ekaterinburg, Russia, and colleagues have synthesized MoS5 and WS5 by a low-temperature solution method from the corresponding carbonyls M(CO)6 (M = Mo, W) and elemental sulfur. The compounds were characterized using a wide range of analytical techniques, including differential scanning calorimetry (DSC), X‐ray diffraction (XRD), energy‐dispersive X‐ray spectroscopy (EDX), thermogravimetric analysis (TGA), X‐ray photoelectron spectroscopy (XPS), scanning electron microscopy (SEM), and infrared (IR) spectroscopy.
The team found that the chain structures of these compounds feature flexible bridging –S–S– groups. These groups make the chains very flexible, which causes their assembly into globular shapes and prevents crystallization. As a result, these sulfur-rich compounds are remarkably different from most of the crystalline chalcogenides and behave rather like conventional polymers. The researchers showed, for example, that sulfur-rich sulfides can be used as cathodes for lithium-ion batteries. The cathodes show increased capacites.
- Revealing the Flexible 1D Primary and Globular Secondary Structures of Sulfur‐Rich Amorphous Transition Metal Polysulfides,
Sofya B. Artemkina, Andrey N. Enyashin, Anastassiia A. Poltarak, Anastasiya D. Fedorenko, Anna A. Makarova, Pavel A. Poltarak, Eon‐Ji Shin, Seong‐Ju Hwang, Sung‐Jin Kim, Ekaterina D. Grayfer, Vladimir E. Fedorov,
ChemNanoMat 2019.
https://doi.org/10.1002/cnma.201900526