Chromium is a poisonous heavy metal that is used in large quantities in different industries. Its hexavalent form Cr(VI) is toxic, carcinogenic, and harmful for the environment. Removal of Cr from industrial wastewaters is, therefore, required. However, conventional methods such as precipitation, adsorption, ion exchange, or electrochemical methods are costly, difficult to use, and not environmentally friendly. Membrane processes such as reverse osmosis and nanofiltration, on the other hand, often have issues with membrane fouling and high hydraulic pressures.
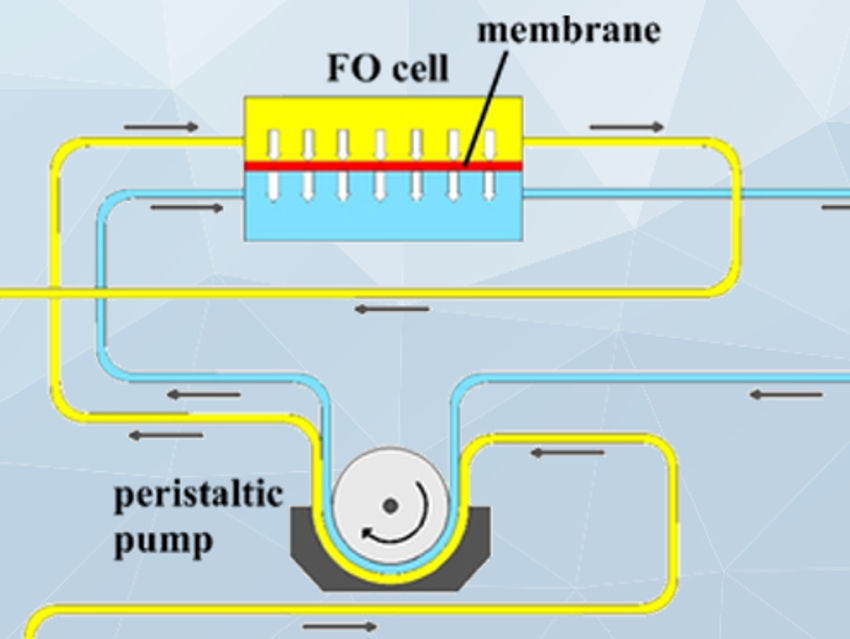
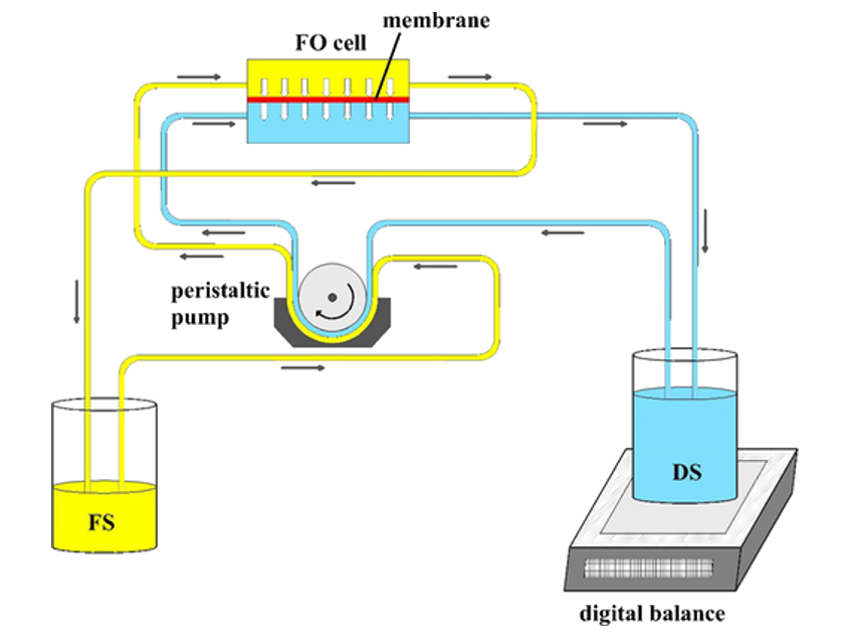
Hamzeh Ali Jamali, Qazvin University of Medical Sciences, Iran, and colleagues have used forward osmosis for chromium removal. Forward osmosis is a green membrane technology that is cheaper, requires less energy, and shows less membrane fouling than other approaches. The feed solution (FS, schematic setup pictured below) is separated from the draw solution (DS) by a semi-permeable membrane, in this case, an aquaporin-based biomimetic membrane. The osmotic pressure between the two sides then drives the permeation of water to the side of the higher concentration, ideally from the feed to the draw side.
As expected, the water flux is greater at higher DS concentrations. A less concentrated feed leads to a more efficient Cr rejection. Longer process times can increase the Cr rejection efficiency. However, one also has to consider the reverse solute flux (RSF), a measure for the solutes from the DS traveling in direction of the FS, i.e., the wrong way. This is not affected by the FS concentration, but the RSF grows with increasing DS concentration. Carefully optimized conditions of the forward osmosis process finally allowed a Cr rejection ratio of nearly 98 %. This is promising for future applications in the clean-up of heavy-metal-containing wastewaters.
- Optimization of the Forward Osmosis Process Using Aquaporin Membranes in Chromium Removal,
Zohreh Naghdali, Soleyman Sahebi, Milad Mousazadeh, Hamzeh Ali Jamali,
Chem. Eng. Technol. 2019.
https://doi.org/10.1002/ceat.201900381