Pathogens such as bacteria, viruses, or fungi can not only infect humans and animals, but also plants and other fungi. The ability to disable chemical defense mechanisms is often very important for the successful invasion of pathogens. Such counterdefense mechanisms have been studied for a long time in the case of fungal pathogens that infect important crops, such as rice, maize or potatoes. In contrast, the defense mechanisms of basidiomycetes fungi—a group of higher fungi that includes, e.g., mushrooms—against pathogenic parasitic fungi are still widely unknown. The same is true for the counterdefense mechanisms of the parasites.
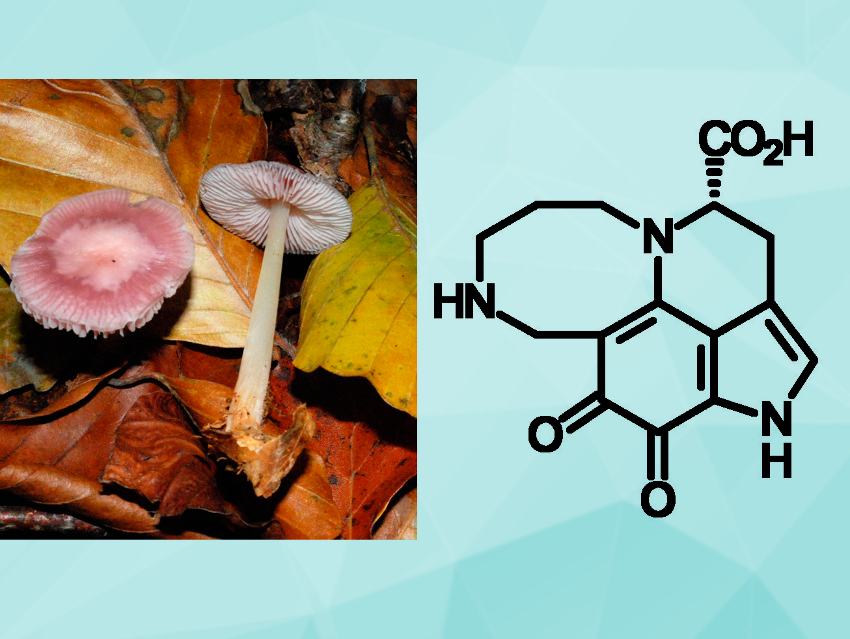
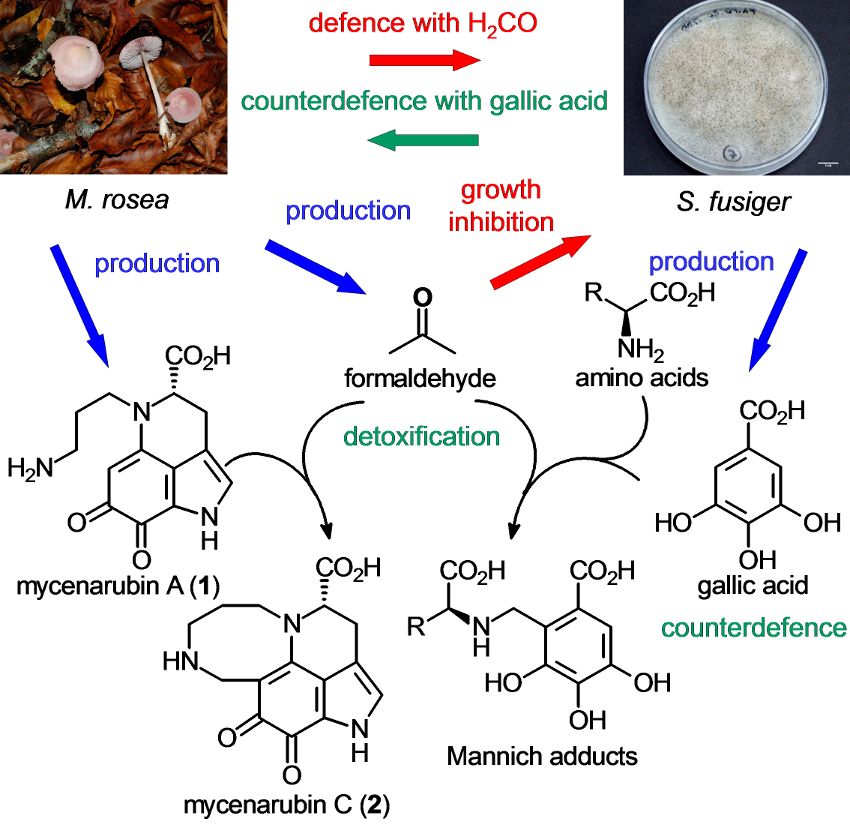
Peter Spiteller, University of Bremen, Germany, and colleagues have investigated the chemical composition of the fruiting bodies of the basidiomycete Mycena rosea (pictured above on the left). The team isolated mycenarubin C, a previously unknown red pyrroloquinoline alkaloid (pictured above on the right). They investigated the biosynthesis of this compound and found that it can be formed from mycenarubin A, the major pyrroloquinoline alkaloid in the fungus, by a reaction with formaldehyde.
Formaldehyde is a toxic compound. This led the researchers to the detection of a chemical defense mechanism in M. rosea: The fungus uses the formaldehyde to inhibit the growth of the mycoparasitic fungus Spinellus fusiger. As a “countermeasure”, S. fusiger can produce large amounts of gallic acid, presumably to inactivate the toxic formaldehyde by the formation of Mannich adducts with amino acids and gallic acid (pictured below).
- Formaldehyde as a Chemical Defence Agent of Fruiting Bodies of Mycena rosea and its Role in the Generation of the Alkaloid Mycenarubin C,
Rieke Himstedt, Silke Wagner, Robert J. R. Jaeger, Michèle-L. Lieunang Watat, Jana Backenköhler, Zeljka Rupcic, Marc Stadler, Peter Spiteller,
ChemBioChem 2020.
https://doi.org/10.1002/cbic.201900733