Fossil discoveries in the Riversleigh World Heritage Area in Australia are crucial to understanding the evolution and effects of climate change on life during the Cenozoic Era. Most deposits at Riversleigh have been found in calcite-rich rocks or sediments, and the fossils are easily removed and their surfaces cleaned using only diluted acetic acid. However, other, newly discovered fossil deposits are more iron-rich, and the physical extraction of the fossils from iron-rich cements is time-consuming and can damage the fossil surfaces. Conventional chemical extraction methods have either been ineffective in dissolving the iron minerals or operate in pH ranges where damage occurs to fossilized bones or teeth.
M. Clara F. Magalhães, University of New South Wales, Sydney, Australia, and Universidade de Aveiro, Portugal, and colleagues have used siderophores (Greek for “iron carriers”) to solve this problem. Siderophores are molecules that form stable chelates and can sequester iron(III) ions, resulting in the dissolution of otherwise low-solubility minerals such as hematite (Fe2O3) or goethite (FeO(OH)). The siderophores acetohydroxamic acid (aHA), salicylaldehyde isonicotinoyl hydrazone (SIH), and pyridoxal isonicotinoyl hydrazone (PIH) (pictured below) can dissolve such iron(III) minerals at pH 9–11 from host matrices and cements encasing fossils without causing noticeable damage to bones or teeth (example pictured above).
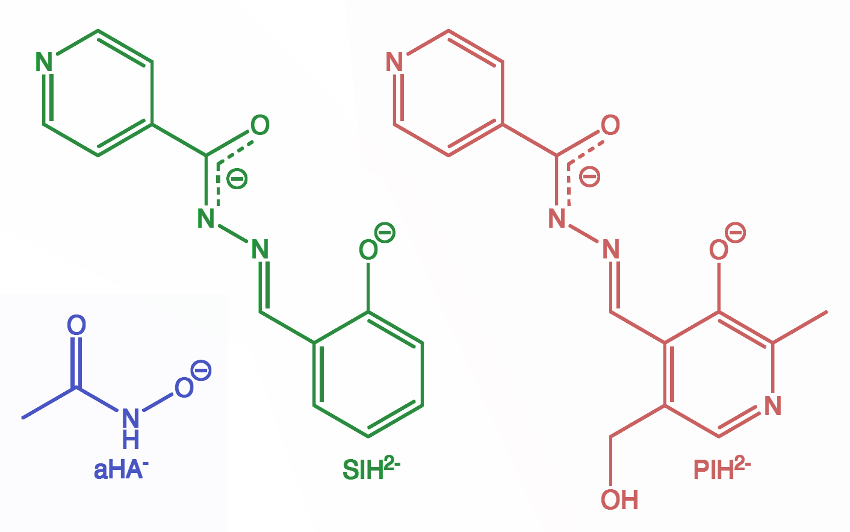
All three siderophores were capable of removing iron(III) hydroxide oxides from the surface of fossils and the encasing matrix without significant damage. However, aHA proved the most effective. The team attributes this to the simplicity of its structure compared with SIH and PIH and its greater solubility across the pH ranges required to preserve bone material.
- Siderophore-Assisted Dissolution of Iron(III) Hydroxide Oxides from Iron-Rich Fossil Matrices,
Justin da Costa, David R. Cohen, M. Clara Ferreira Magalhães, D. Brynn Hibbert, Michael Archer, Troy J. Myers, Suzanne J. Hand,
ChemPlusChem 2020.
https://doi.org/10.1002/cplu.202000407




