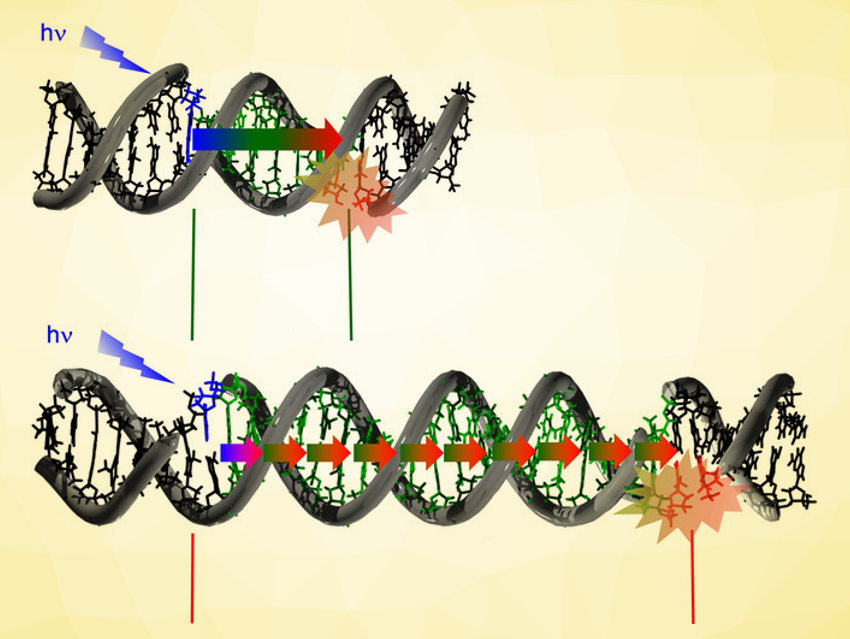
Exposure to ultraviolet (UV) light from the sun can affect the integrity of genetic material. Cyclobutane pyrimidine dimers (CPDs) are the main DNA lesion caused by UV light. These lesions represent a common cause of skin cancer. However, how far energy migrates in DNA and forms CPDs is an open question.
Hans-Achim Wagenknecht and colleagues, Karlsruhe Institute of Technology (KIT), Germany, have developed a synthetic DNA architecture that can be used to address this question. The team used a xanthone as an artificial nucleotide that can be selectively excited by UV-A light and, thus, allows site‐selective photoenergy injection into DNA (pictured above in blue). As a designated photodamage site, they used two adjacent thymidines (pyrimidine-containing nucleosides, pictured above in red) with no phosphodiester bond between them. Both sites can be placed in well-defined distances along a DNA strand. When the energy migrates through DNA and forms CPDs, the resulting cyclobutane links two oligonucleotides together, which allowed the team to probe the damage using gel electrophoretic analysis.
The researchers found that there is a turning point between short-range and long-range energy migration at a distance of 25 Å. Below this distance, there are high CPD yields. Remarkably, significant amounts of photodamage still occurred over long distances up to 105 Å (or 30 base pairs). This observation shows that DNA photodamage does not only occur locally at the site of excitation.
- How Far Does Energy Migrate in DNA and Cause Damage? Evidence for Long‐Range Photodamage to DNA,
Arthur Kuhlmann, Larissa Bihr, Hans‐Achim Wagenknecht,
Angew. Chem. Int. Ed. 2020.
https://doi.org/10.1002/anie.202009216