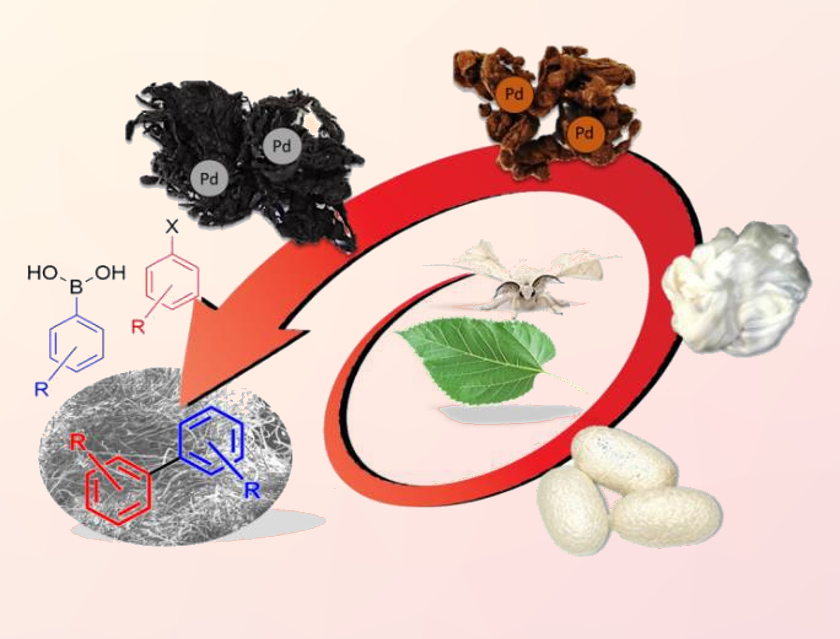
Palladium catalysts supported on natural biopolymers, such as cellulose, chitosan, alginate, or wool, can be used for sustainable Suzuki-Miyaura cross-coupling reactions. Supported Pd catalysts such as these can be easily separated from the reaction mixture and reused. However, a need for high catalytic loading and severe metal leaching are problematic for these protocols.
Gianluca M. Farinola, University of Bari Aldo Moro, Italy, and colleagues have used silk fibroin (SF), commonly available from cocoons of the domesticated silkworm (Bombyx mori), as a convenient biological support for a Pd catalyst in Suzuki-Miyaura reactions. To prepare the catalyst, SF was soaked for 15 min in a boiling aqueous solution of PdCl2 to obtain Pd(II)-impregnated fibers.
Using the resulting supported catalyst, coupling reactions of aryl iodides, bromides, and chlorides with aryl boronic acids were performed in a water/ethanol mixture under atmospheric conditions. The reactions proceeded rapidly with low metal loading (0.38 mol%) and gave high yields. Compared with other biopolymer-supported palladium catalysts for the Suzuki-Miyaura coupling, the SF-supported catalyst shows improved recyclability, with up to 19 cycles without loss of catalytic activity. The protocol allows fast, scalable, versatile, and low-cost access to biaryl compounds.
- Palladium supported on silk fibroin for Suzuki‐Miyaura cross‐coupling reactions,
Giorgio Rizzo, Gianluigi Albano, Marco Lo Presti, Antonella Milella, Fiorenzo G. Omenetto, Gianluca M. Farinola,
Eur. J. Org. Chem. 2020.
https://doi.org/10.1002/ejoc.202001120