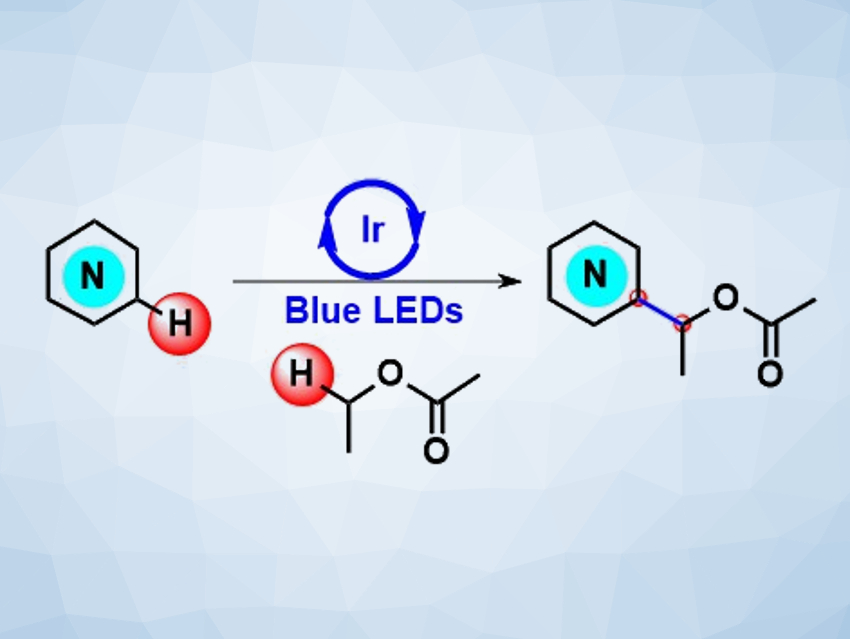
The direct functionalization of nitrogen-containing heterocycles is of great interest to the chemical industry, since it avoids multistep synthetic routes and pre-functionalization of the reactants. Methods based on visible-light-induced mild radical generation, for example, can enable key transformations, such as cross-dehydrogenative C–C coupling reactions.
However, the concept of using common organic solvents as reactants, and—more importantly—as radical precursors, is not frequently used in the design of chemical reactions. In general, solvents with a C(sp3)-H bond adjacent to an oxygen atom (e.g., alcohols or ethers) can be cleaved via a hydrogen atom transfer (HAT) process, and the corresponding radical can react with heteroarenes directly. Protected alcohols, such as ethyl acetate, have rarely been studied as a radical source in organic reactions so far.
Márió Gyuris, Servier Research Institute of Medicinal Chemistry, Budapest, Hungary, and colleagues have developed a visible-light photoredox direct α-acyloxyalkylation reaction of N-heterocycles (pictured) that uses ethyl acetate as a low-cost, non-conventional radical source. The team used (Ir[dF(CF3)ppy]2(dtbbpy))PF6 as a photocatalyst (ppy = 2-phenylpyridine, dtbbpy = 4,4′-bis(tert-butyl)-2,2′-bipyridine), (1S)‐(+)‐10‐camphorsulfonic acid (CSA) as the acid, and tert‐butyl peroxybenzoate (TBPB) as a radical initiator. The reactions were performed under blue LED light.
The cross-dehydrogenative coupling proceeds via the generation of the 1-acetoxyethan-1-yl radicals. The reaction provides access to a structurally diverse set of α-acyloxyalkylated N-heteroaromatics, which can be pharmaceutically relevant intermediates. The team also developed a procedure for the preparation of the model compound 1-(4-methylquinolin-2-yl)ethyl acetate on a multigram scale.
- Visible‐Light Photoredox Alkylation of Heteroaromatic Bases Using Ethyl Acetate as Alkylating Agent,
Nándor Győrfi, András Kotschy, Márió Gyuris,
Eur. J. Org. Chem. 2020.
https://doi.org/10.1002/ejoc.202001113