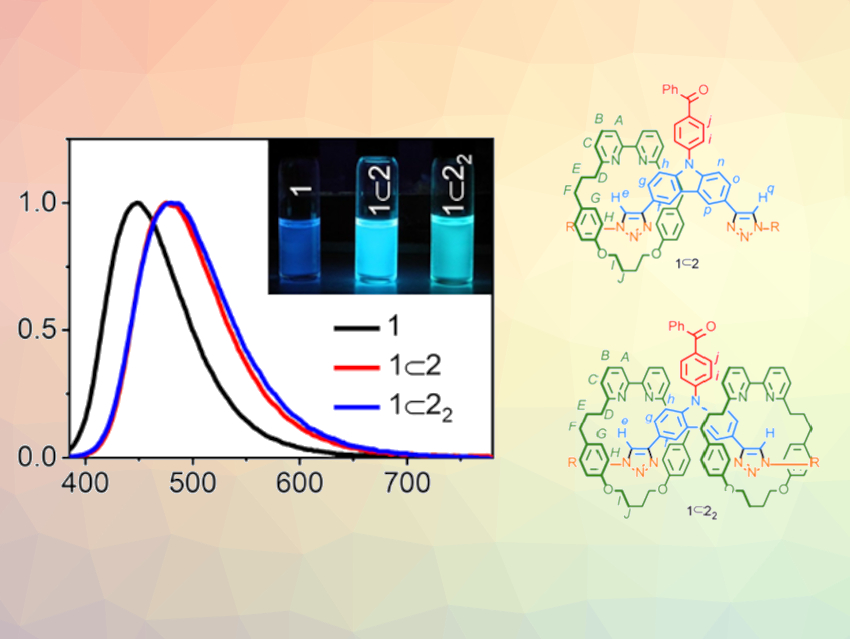
Thermally activated delayed fluorescence (TADF) emitters are promising for applications in organic optoelectronics such as organic light-emitting diodes (OLEDs). They enable a high device efficiency by harvesting both singlet and triplet excitons (quasiparticles consisting of an electron and an electron hole). Usually, molecules consisting of donor and acceptor groups are used as TADF emitters. The efficiency of a TADF process depends on the degree to which the electron density of the donor and the acceptor fragments overlap.
Ifor D. W. Samuel, Eli Zysman‐Colman, University of St Andrews, UK, Thomas J. Penfold, Newcastle University, Newcastle upon Tyne, UK, Stephen M. Goldup, University of Southampton, UK, and colleagues have found that the mechanical bond in a rotaxane can be used to fine-tune the photophysical properties of a TADF emitter. The team synthesized two interlocked TADF emitters: [2]rotaxane 1⊂2 and [3]rotaxane 1⊂22 (pictured below).
The rotaxanes were synthesized using a copper-mediated azide‐alkyne cycloaddition between a benzophenone-substituted carbazole bis‐alkyne unit (benzophenone pictured in red, carbazole unit pictured in blue) and a bulky benzylic azide (pictured in yellow) in the presence of macrocycle 2 (pictured in green).
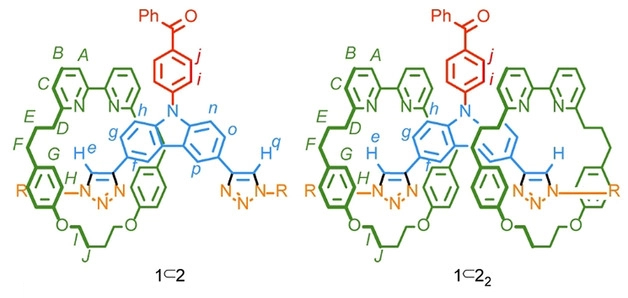
Compared with the non-interlocked “axle” 1 (pictured in red, blue, and yellow), the introduction of the mechanical bonds results in increased efficiency of light emission, easier conversion of triplets to singlets, and improved photostability. Computational simulations and X-ray crystallography results suggest this subtle tuning is due to the mechanical bond enforcing weak interactions between the axle and the macrocycle. This work could provide a new approach for optimizing TADF emitters and increasing device efficiency.
- Using the Mechanical Bond to Tune the Performance of a Thermally Activated Delayed Fluorescence Emitter,
Pachaiyappan Rajamalli, Federica Rizzi, Wenbo Li, Michael Jinks, Abhishek Kumar Gupta, Beth Laidlaw, Ifor Samuel, Thomas Penfold, Steve Goldup, Eli Zysman-Colman,
Angew. Chem. Int. Ed. 2021.
https://doi.org/10.1002/anie.202101870