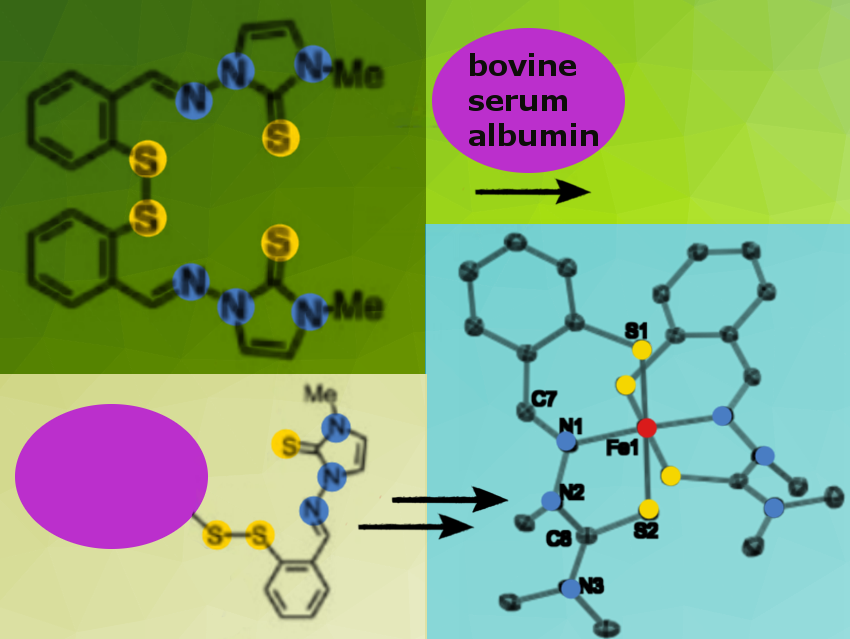
Cancer cells show a higher demand for iron to support higher proliferation rates. Several iron-binding compounds, such as thiosemicarbazone chelators, exploit this vulnerability of malignant cells and are currently being investigated as anticancer therapeutic candidates. Disulfide-based prochelators target intracellular iron pools because they are activated for iron coordination upon cellular uptake and reductive activation in the presence of elevated levels of glutathione and other intracellular reductants.
Elisa Tomat, Michael Marty and their research teams, University of Arizona, Tucson, USA, synthesized disulfide-based prochelators with alkylated thiosemicarbazone and imidazole-2-thione binding units (example pictured in dark green). The corresponding thiols form iron(III) complexes under aerobic conditions (pictured blue) as the researchers confirmed by crystallography and electron paramagnetic resonance (EPR) experiments.
In the presence of serum albumin, which is a major component of cell growth media and the most abundant protein in human blood, the new prochelators form conjugation products (pictured yellow). A thiol-disulfide exchange takes place involving a reduced cysteine residue on the protein. Native mass spectrometry experiments showed clear covalent modification of the protein upon 24-hour incubations.
In a panel of breast, ovarian, and colorectal cancer cell lines, the constructs present antiproliferative activities at sub-micromolar concentrations — the best activity profiles within their class of disulfide-based prochelators. In contrast, normal lung fibroblasts are far less susceptible to the compounds, and antiproliferative activities are up to two orders of magnitude lower.
The new prochelators, therefore, combine a promising toxicity profile with albumin bioconjugation, which could confer enhanced lifetime in blood circulation and tumor accumulation. The researchers look forward to learning more about the specific intracellular effects of the iron-binding systems as well as their anticancer activity in vivo.
- Albumin conjugates of thiosemicarbazone and imidazoleâ€2â€thione prochelators: Iron coordination and antiproliferative activity,
Yu-Shien Sung, Wangbin Wu, Megan A. Ewbank, Rachel D. Utterback, Michael T. Marty, Elisa Tomat,
ChemMedChem 2021.
https://doi.org/10.1002/cmdc.202100278