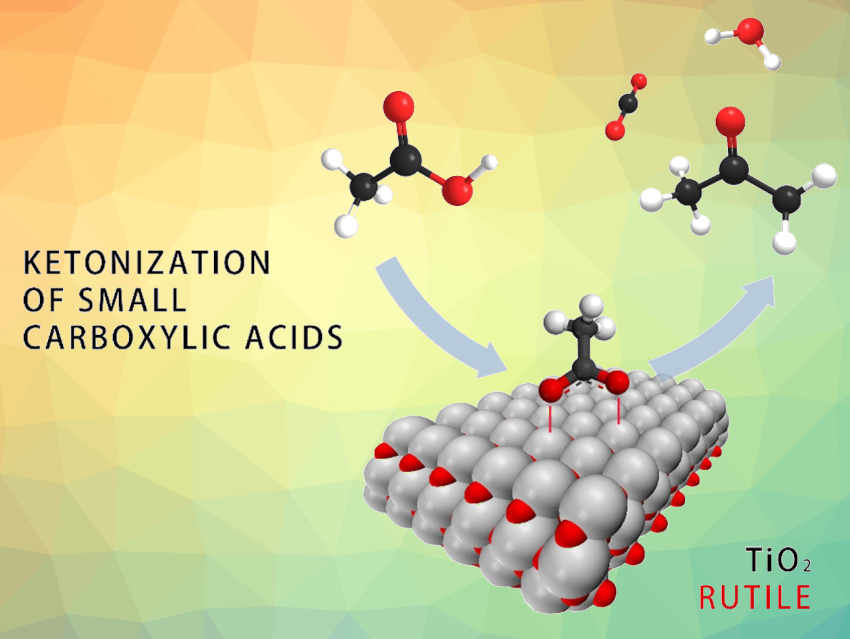
Sustainability in the production of chemicals can be improved by “closing the carbon loop”, e.g., by using carbon-containing waste as a resource. The carbon contained in aqueous wastewater streams can, for example, be captured by microorganisms and accumulated in the form of short-chain volatile fatty acids (VFAs). These VFAs can serve as versatile chemical building blocks. They can, e.g., be transformed using a catalytic ketonization (pictured) to give precursors for fuel components, lubricants, or alkylated aromatics. This ketonization reaction is typically catalyzed by metal oxides such as TiO2. However, the reaction mechanism and the nature of active surface species in this type of reaction are not fully understood.
Pieter C. A. Bruijnincx, Utrecht University, The Netherlands, and colleagues have compared the catalytic activity of different TiO2 polymorphs, i.e., anatase, rutile, and brookite. The team used propionic acid as a model substrate. The gas-phase ketonization of this compound was investigated at temperatures of 325 °C, 375 °C, and 425 °C, using equal weights of the three TiO2 polymorphs. The team found rutile to be the most active and selective catalyst over the studied range of temperatures. The second most effective polymorph was brookite, while phase-pure anatase performed poorly.
Operando Fourier-transform infrared (FT-IR) spectroscopy results allowed the researchers to link the catalytic performance to the mode of adsorption of the acid on the TiO2 surface. Bidentate carboxylates were the most abundant species on the highly active rutile and brookite surfaces, while monodentate and bidentate carboxylates were present in similar amounts on the anatase surface. Overall, the work provides new insight into the mechanism of ketonization and could contribute to the further development of the key catalytic step in this waste-to-value approach.
- Crystal Phase Effects on the Gas‐Phase Ketonization of Small Carboxylic Acids over TiO2 Catalysts,
Egor Fufachev, Bert Weckhuysen, Pieter Bruijnincx,
ChemSusChem 2021.
https://doi.org/10.1002/cssc.202100721