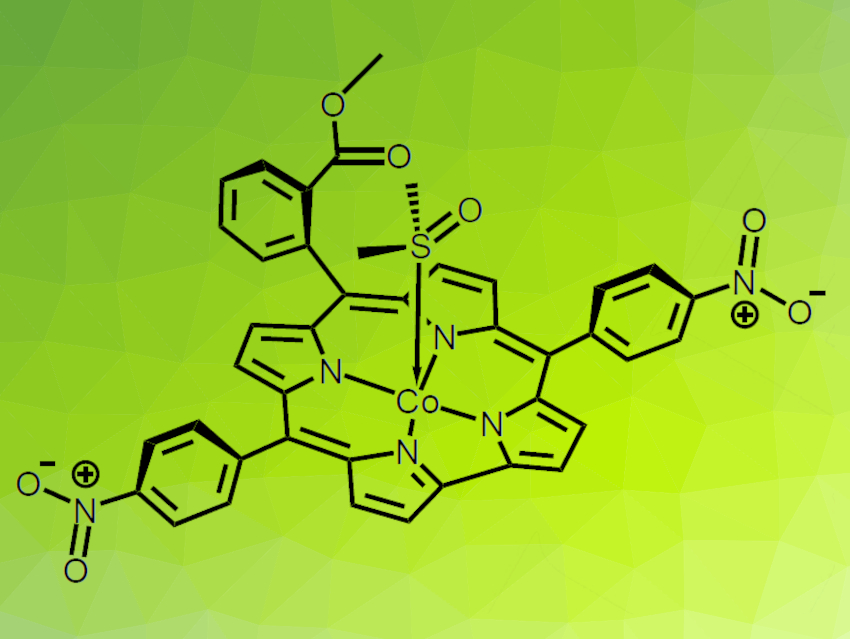
Metallocorroles are coordination compounds related to the heme group, which transports O2 in blood, and similar to the essential vitamin B12. They can be used in important catalytic applications such as H2 evolution, CO2 reduction, and water oxidation. A good understanding of their properties and the effects of structural variations is important for these applications.
Biprajit Sarkar, University of Stuttgart, Germany, Nicolás I. Neuman, University of Stuttgart and Instituto de Desarrollo Tecnológico para la Industria Química (INTEC), Santa Fe, Argentina, and colleagues have reported the first structure of a cobalt metallocorrole with a DMSO apical ligand (pictured), a low-spin Co(III) complex of 5,15-bis(4-nitrophenyl)-10-(2-methylcarboxyphenyl)corrole. The complex was prepared from the free base corrole and Co(OAc)2·4 H2O in DMSO. The product was characterized using cyclic voltammetry, infrared (IR), UV-Vis, NMR, and electron paramagnetic resonance (EPR) spectroscopy, as well as X-ray crystallography.
The ligand binds to cobalt through a sulfur atom, which influences the electronic, magnetic, and redox properties of the complex due to a large σ-donor effect of the S atom. According to the researchers, DMSO is an ideal ligand for catalysis, as it binds strongly to the metal, is oxidatively stable, and leaves an empty site on the metal where a substrate can bind and react. The insights obtained in this work could be helpful for further investigations of the applications of DMSO-ligated cobalt corroles, especially in the field of energy-related catalysis. The results also indicate interesting possibilities for the immobilization of cobalt(III) corroles on sulfoxide-functionalized surfaces.
- Synthesis and Characterization of a Cobalt(III) corrole with an S‐bound DMSO ligand,
Nicolás I. Neuman, Arijit Singha Hazari, Julia Beerhues, Fabio Doctorovich, Santiago E. Vaillard, Biprajit Sarkar,
Eur. J. Inorg. Chem. 2021.
https://doi.org/10.1002/ejic.202100524This article is part of the special collection “Inorganic Chemistry in Latin America“.