“Flow or batch?” ask K. F. Jensen et al. in their Review on flow reactors in synthesis, and discuss the impact on synthetic chemistry on all scales—from the laboratory to full production. In a Minireview, J. Barluenga and C. Valdés describe the use of tosylhydrazones in palladium-catalyzed and metal-free cross-coupling reactions. L. R. Giam and C. A. Mirkin contribute an Essay on cantilever-free scanning probe molecular printing. The Highlights deal with the enzyme toolbox for biocatalysis (J. E. Vick and C. Schmidt-Dannert) and dehydrogenative cross-coupling reactions of heteroarenes (X. Bugaut and F. Glorius). In an Editorial, H. Wild et al. point out the importance of chemistry for the future of the pharma industry.
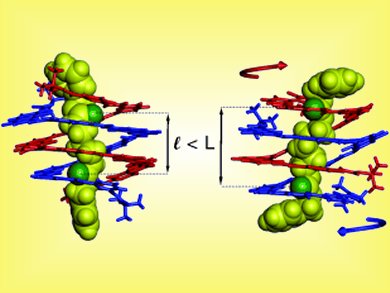
In the Communications section, I. Huc et al. present an antiparallel double-helical aromatic oligoamide foldamer that binds to a series of rodlike guests of various lengths upon the winding of the duplex around the guests (see scheme). C. A. Russell et al. show that gold(I) cations bind to only one double bond of a 1,3-diene unit, but can glide over the C4 backbone on a low-activation-energy pathway. M. Shibasaki et al. describe a facile path to enantiomerically enriched 3-hydroxy-2-oxindoles. C. Hoppmann et al. report on the light-controlled contraction of muscle fibers.