Drugs that are selective for cyclooxygenase-2 (COX-2) over COX-1 carry a lowered risk of gastrointestinal irritation. Merck’s rofecoxib is an example of a selective COX-2 inhibitor that was effective at treating chronic pain and inflammation without the side effects of stomach upset. However, this drug was pulled from the market roughly five years after its launch, as its chronic use was found to increase the risk of heart attack and stroke; this was caused by decreased levels of the beneficial vasodilatory and anti-aggregatory prostacyclin, and increased amounts of thromboxane A2, which induces vasoconstriction and promotes platelet aggregation.
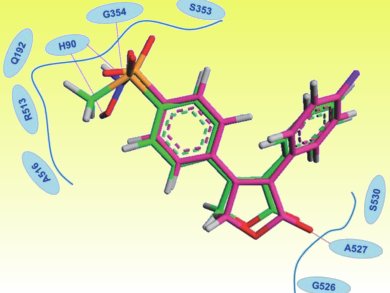
A team led by Edward Knaus, University of Alberta, Canada, has discovered a suitable way to circumvent the adverse elevation of blood pressure and increased prevalence of blood clots: they replaced the SO2Me substituent in rofecoxib with a nitric oxide (NO) donor sulfohydroxamic acid (SO2NHOH) COX-2 pharmacophore. The idea being that NO is a vasorelaxant and an inhibitor of platelet aggregation. The group synthesized a group of stable and readily isolated candidate lead compounds (pictured).
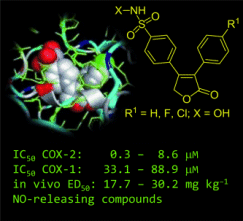
Biological studies revealed the chloro-substituted compound stands out as a particularly potent and selective inhibitor of COX-2. It is also an effective NO donor at physiological pH, and has significant anti-inflammatory activity.
- Rofecoxib Analogues Possessing a Nitric Oxide Donor Sulfohydroxamic Acid (SO2NHOH) Cyclooxygenase-2 Pharmacophore: Synthesis, Molecular Modeling, and Biological Evaluation as Anti-inflammatory Agents
A. Bhardwaj, Z. Huang, J. Kaur, E. E. Knaus,
ChemMedChem 2012, 7(01).
DOI: 10.1002/cmdc.201100393