Dietmar Stalke and colleagues, Georg August University, Göttingen, Germany, have made two unusual white phosphorus (P4)-based compounds (pictured). The acyclic P4 chain and silicon-phosphorus cage were formed by activation of P4 by silylenes. This is the first example of an acyclic Si–P chain that contains 6π electrons. Chain or cage formation was dictated by the substitution on the silylenes, with the three-coordinate [PhC(NtBu)2SiN(SiMe3)2] giving the chain, while the two-coordinate [(Me5C5)SiN(SiMe3)2] gave the cage structure.
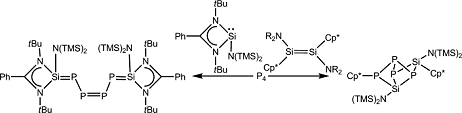
This gives a mild route to P4 activation, which is of industrial interest for the more facile production of organophosphorus derivatives. The resulting Si–P compounds can be used as electron-rich chelating ligands for transition metals and may find application in metal-mediated catalytic process. They may also have commercial application as precursors for semiconducting materials.
- A P4 Chain and Cage from Silylene-Activated White Phosphorus
S. Khan, R. Michel, S. S. Sen, H. W. Roesky, D. Stalke,
Angew. Chem. Int. Ed. 2011.
DOI: 10.1002/anie.201105610