The catalytic hydrogenation of polar bonds, in particular organic carbonyl groups, provides an environmentally benign approach to synthetically important building blocks, such as alcohols and amines. Urea derivatives are the most challenging polar carbonyl groups to hydrogenate. Catalytic hydrogenation of urea derivatives has never been reported. Alkyl urea compounds have been used as solvent in hydrogenation reactions.
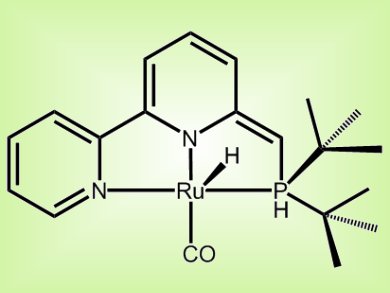
David Milstein and co-workers, Weizmann Institute of Science, Israel, report the first example of catalytic hydrogenation of urea derivatives to the corresponding amines and methanol. The reaction is catalyzed by a bipyridine-based tridentate PNN Ru(II) pincer complex (pictured) and proceeds under mild, neutral conditions using 13.6 atm of H2.
As alkyl and aryl urea derivatives are readily obtained from CO2 and amines, their hydrogenation offers an environmentally benign, mild, atom economical approach to the indirect transformation of CO2 to methanol, which is of intense current interest with regard to hydrogen storage and “methanol economy”.
- Unprecedented Catalytic Hydrogenation of Urea Derivatives to Amines and Methanol
E. Balaraman, Y. Ben-David, D. Milstein,
Angew. Chem. Int. Ed. 2011.
DOI: 10.1002/anie.201106612