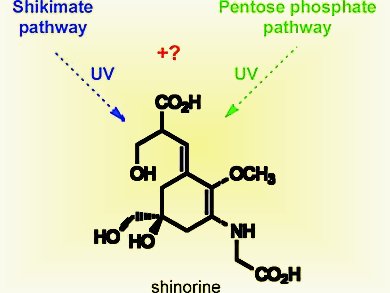
Cyanobacteria are phototrophic prokaryotes, they obtain their energy through photosynthesis. They have evolved ultraviolet-screening mycosporine-like amino acids (MAAs) to ease the harmful effects of their obligatory exposure to solar radiation in their shallow water habitats.
Multiple lines of evidence provide that MAAs are biosynthesised via the shikimic acid pathway, but a different MAA biosynthetic pathway was recently reported. Biosynthesis of the MAA shinorine in Anabaena variabilis (ATCC 29413) and Nostoc punctiforme (ATCC 29133) via the intermediates 4-deoxygadusol and mycosporine-glycine was found to proceed from the sedoheptulose 7-phosphate (SH 7-P) precursor of the pentose phosphate pathway, rather than 3-deoxy-d-arabinoheptulosonate 7-phosphate (DAHP) of the shikimic acid pathway (scheme).
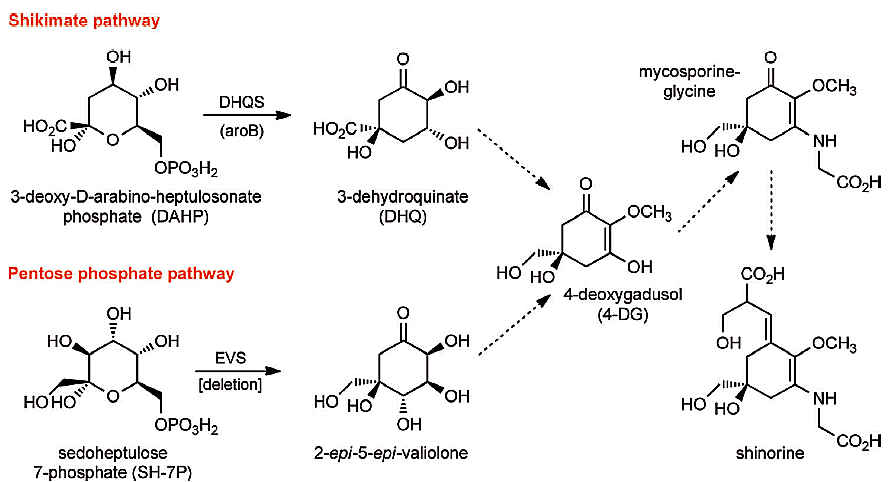
The cyanobacterial SH 7-P cyclase, 2-epi-5-epivaliolone epivaliolone synthase (EVS), is a key biosynthetic enzyme of this pathway and is strikingly similar to the DAHP cyclase, 3-dehydroquinate synthase (DHQS), of the shikimic acid pathway. Both are members of the same superfamily of enzymes and both lead to the convergent MAA precursor, 4-deoxygadusol.
Paul F. Long, King’s College London, UK, and colleagues examine the effect on MAA biosynthesis of deleting the EVS gene in the cyanobacterium Anabaena variabilis. This shows that Anabaena variabilis does not biosynthesise shinorine exclusively through the pentose phosphate pathway.
- Redundant Pathways of Sunscreen Biosynthesis in a Cyanobacterium,
Edward Spence, Walter C. Dunlap, J. Malcolm Shick, Paul F. Long,
ChemBioChem 2012.
DOI: 10.1002/cbic.201100737