A variety of molecular machines that are capable of changing their state in response to external chemical, electrochemical, and photochemical stimuli have been prepared lately. Among these are self-locking systems which have been used for phototriggered catalysis. Modulation or even clear cut ON/OFF regulation of catalytic activity by chemical stimuli has not been reported yet.
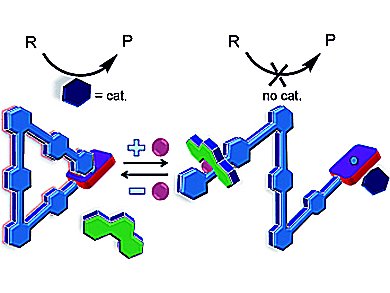
Michael Schmittel, Soumen De, and Susnata Pramanik, University of Siegen, Germany, report on the synthesis of a selflocking system. This nanoswitch uses chemical inputs to turn an organocatalytic Knoevenagel reaction on and off (see scheme: R = reactant, P = product).
In the locked state, piperidine (dark blue) is not coordinatively trapped at the porphyrin docking station of the self-locking system. As a consequence, piperidine is catalytically active in a Knoevenagel reaction (ON).
In the presence of Cu+(pink) and phenanthroline (green) as external stimuli, the self-locking system is unlocked, thus allowing for strong coordination of piperidine (dark blue) to the zinc porphyrin docking station. In this state, catalysis stops (OFF).
Upon addition of phenanthroline (green) to the piperidine···[Cu(self-locking system)(phenanthroline)]+, the complex breaks down to release the catalytically active piperidine (dark blue). The process can run reversibly over three cycles without loss of activity.
- Reversible ON/OFF Nanoswitch for Organocatalysis: Mimicking the Locking and Unlocking Operation of CaMKII,
Michael Schmittel, Soumen De, Susnata Pramanik,
Angew. Chem. Int. Ed. 2012.
DOI: 10.1002/anie.201108089