The use of functionalized nucleic acids for applications in nanotechnology has emerged as an independent area of research with implications in the fields of diagnostics, electronics, and material sciences.
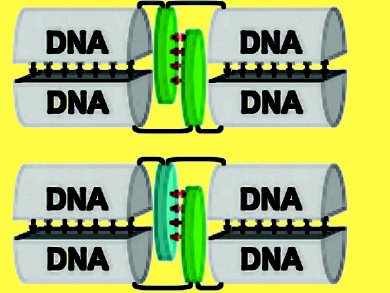
Florian Garo and Robert Häner, University of Bern, Switzerland, synthesized two types of 2,1,3-benzothiadiazole-based phosphoramidite building blocks. The building blocks were integrated into oligodeoxynucleotides through alkynyl or carboxamide linkers (W and V). Both building blocks were found to support duplex stability in hybrids containing a 2,1,3-benzothiadiazole (BTD) unit in each of the strands in opposite positions through interstrand stacking interactions. This stabilization is attributed to the high polarization in the BTD molecules caused by the electron-withdrawing thiadiazole and the favorable π–π stacking interactions resulting therefrom. Placement opposite to a natural base, however, resulted in considerable destabilization.
The bis(alkynyl)-derived BTD W exhibited a fluorescence behavior that was strongly dependent on the neighboring nucleobases. The quenching effect decreased in the order G >> A ≥ T ≥ C and correlated very well with the calculated free energies for charge separation (ΔGCS) through photoinduced electron transfer.
Fluorescence of W is essentially completely quenched when it is placed against the bis(carboxamide)-derived BTD V.
The described BTD-based compounds W and V represent valuable building blocks for the construction of highly ordered, DNA-based materials with special optical and electronic properties.
- 2,1,3-Benzothiadiazole-Modified DNA,
Florian Garo, Robert Häner,
Eur. J. Org. Chem. 2012.
DOI: 10.1002/ejoc.201200231