‘Bath salts’ law tries to keep pace with designer drug makers [1]
Father Whose Son Took His Life After Using K2 to Speak in West Des Moines Monday [2]
US Senate Hearing on the Dangers of Synthetic Drugs [3]
Headlines from Phoenix, Arizona, USA, to Frankfurt, Germany, repeat the same story – lives destroyed by increasingly powerful and sophisticated drugs and law enforcement trying to keep up with a dizzying array of new “designer” drugs and drug combinations engineered to be difficult to detect or characterize, and often to skirt existing drug laws. The International Narcotics Control Board (INCB) has described the proliferation of designer drugs as escalating out of control [4].
Traditionally, labs have relied on standards and libraries to be able to identify compounds, but, with designer drugs, standards may not be available. As a result, labs face significant challenges in being able identify these compounds, their precursors and metabolites – doing so rapidly and accurately to meet the needs of healthcare providers and law enforcement is nearly unmanageable. Even with certified samples from reputable sources, labs face the risk of receiving false or decomposed standards, owing to improper synthesis or transportation. Dr. Peter Rösner, former leader of the section Analysis of Drugs of Abuse and Forensic Toxicology at the Landeskriminalamt in Kiel, Germany, says:
“Any purchased reference standard is to be verified. A check for purity by gas chromatography and mass spectral interpretations is mandatory. If necessary a structural elucidation by spectroscopic methods is performed and a careful interpretation has to be made.”
To help forensic and pathology labs meet these challenges, manufactures are providing a growing arsenal of analytical tools to add to the lab’s armamentarium. High resolution mass spectrometers, in particular, aid in narrowing down the elemental composition of unidentified samples. However, investigators and healthcare providers require more than a short list of chemical candidates. To address this problem head on, Dr. Rösner and his colleagues developed a specialized collection of mass spectral fingerprints that focuses on positively identifying designer drugs. Mass Spectra of Designer Drugs is updated annually, with an online database of newly characterized compounds available in between new editions.
The latest edition, Mass Spectra of Designer Drugs 2012 with more than 16,000 mass spectra, provides labs with a targeted, comprehensive snapshot of illegal drug fingerprints for accurate and rapid identification.
Quality in such a collection is a major concern. Dr. Rösner described the process of selection:
“Before being added to the mass spectral library, the mass spectra go through a stringent verification process, using modern spectroscopic methods. For 15 years most case work samples are elucidated by 1H-NMR, 13C-NMR including two dimensional NMR methods. The molecular weight of the compound is determined by chemical ionization or high resolution mass spectroscopy. Often daughter ion mass spectroscopy is used to get additional structural information. IR spectroscopy is an additional standard. Spectra before this time could not be treated in the same way, because such highly sophisticated methods were not generally available at this time. The early spectra or samples originated from reliable sources or colleagues in crime labs in Germany.”
While some of the compounds are indeed found in larger, heterogeneous collections, such as the Wiley Registry of Mass Spectral Data, or the NIST/EPA/NIH Mass Spectral Library, the spectra in Designer Drugs 2012 cannot be found elsewhere. All of the Designer Drugs spectra have gone through the same quality assurance steps at the DigiLab Software GmbH, and are auditable. Dr Rösner describes the quality assurance process:
“Before a mass spectrum of a compound enters Mass Spectra of Designer Drugs, it is examined by at least three experienced mass spectrometrists skilled in mass spectral interpretations. Any single electron ionization mass spectrum additionally passes through an incorruptible computerized process. This digital evaluation process ends with the calculation of a “Quality Index for Reference Mass Spectra” developed by Professor Fred W. McLafferty. This quality index (QI) is a numerical value between 0 and 1000 and the product of seven quality factors.”
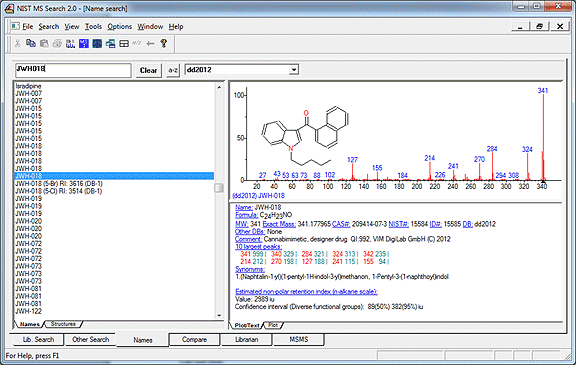
Figure 1. Mass Spectra of Designer Drugs 2012 in NIST MS Search 2.0g. Note the quality index and the annotation “VIM”, indicating that the sample was verified by multiple instruments.
In only rare cases such as ion trap spectra, metabolites, very compact molecules, or an uncommon substance-specific fragmentation, are spectra with a quality index below 900 accepted. In the case of compound synthesis, mass spectroscopic methods (electron ionization mass spectroscopy, chemical ionization, daughter ion mass spectroscopy) are used by default.
For new, emerging designer drugs where no related data or reference standards exist, the challenge is to elucidate the structure of the unknown compound by skilled interpretation of spectral data. Verification by multiple spectroscopic methods and three or more experts improves the reliability of this collection.
While designer drugs continue to proliferate, labs have increasingly powerful tools to fight back in the form of more accurate instruments and more powerful and up-to-date reference libraries.
References
[1] David Carkhuff, Portland Daily Sun, April 03, 2012.
[2] Beth Dalbey, Patch.com, April 11, 2012.
[3] The Dangers of Synthetic Drugs Hearing, Senator Chuck Grassley, YouTube.
[4] International Narcotics Control Board Annual Report, 2 March 2011.