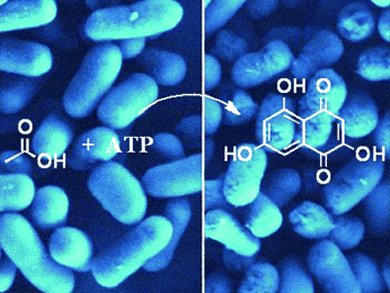
In situ biocatalysis, a known alternative to in vitro and in vivo syntheses, is used to produce the secondary metabolite flaviolin within permeabilized cells of Escherichia coli. A bank of information on the enzymes and metabolic networks in E. coli enables the manipulation of pathway activities and addition.
E. Heinzle and co-workers, Saarland University, Germany, used the detergent Triton X-100 to permeabilize the E. coli cells. Triton X-100 retained the enzymes inside the cells and enabled the entry of the buffer molecules acetate, triclosan, biotin, and the enzyme cofactor ATP hydrate. The permeabilization and reaction required mild conditions of pH 7.8 and T = 30 °C.
The tailored permeabilized E. coli cells produced 0.93 mM flaviolin/flaviolin dimer after a one day incubation at 30 °C, and the overexpression of acetyl-CoA synthase with deletion of competing pathways increased the activity to flaviolin production significantly. The overall yield of 30 % of flaviolin with respect to ATP could owe to ATP sinks in “futile” cells.
Further developments by using bioinformatics and metabolic engineering will help to tailor the catalytic activity, minimize byproducts, and permit growth of the E. coli and, hence, increase in situ enzyme production.
Image: © Wiley-VCH
- Multienzyme Whole-Cell In Situ Biocatalysis for the Production of Flaviolin in Permeabilized Cells of Escherichia coli,
S. Krauser, P. Kiefer, E. Heinzle,
ChemCatChem 2012, 4, 786–788.
DOI: 10.1002/cctc.201100351