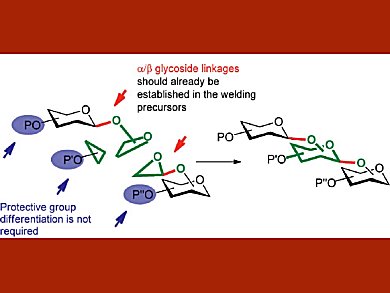
The chemical synthesis of oligosaccharides normally relies on the coupling of separate existing monosaccharide parts through the formation of the glycosidic bond. However, this method often suffers from laborious protection strategies and difficulties with controlling the stereochemistry and position of the glycosidic linkage.
Jatta A. Himanen and Petri M. Pihko University of Jyväskylä, Finland, present a new strategy for the synthesis of di- and trisaccharides based on a de novo saccharide linchpin construction. In this strategy, functionalized monosaccharide building blocks already incorporating the glycosidic linkages are welded together using a Cr-catalyzed hetero-Diels–Alder (HDA) reaction to generate a new monosaccharide unit between them. The highest yields and selectivities in the HDA reaction were obtained by using chiral Schiff base chromium complexes.
For the synthesis of trisaccharide products, acetyl-protected glucose or galactose-derived dienes were fused with monosaccharide-derived aldehydes using chromium catalysts for the HDA reaction. The desired trisaccharide products were obtained in moderate to good yields with excellent stereoselectivity.
The central pyranulose-ring generated in the process possessed an L-cisenulose configuration according to NMR spectroscopy and modeling studies.
- Synthesis of Trisaccharides by Hetero-Diels–Alder Welding of Two Monosaccharide Units,
Jatta A. Himanen, Petri M. Pihko,
Eur. J. Org. Chem. 2012.
DOI: 10.1002/ejoc.201200277