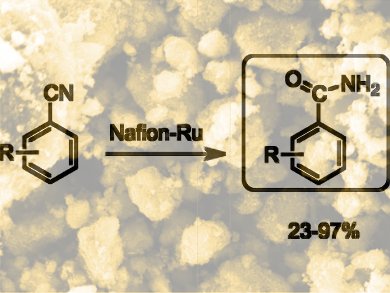
The hydration of nitriles to amides is a relatively simple reaction and several transition-metal catalysts are available for this selective conversion that do not overoxidise the substrates. However, removing the catalysts after the reaction is anything but simple. Metal contamination often remains in the products and large amounts of waste are often generated.
To tackle these problems, Surya Prakash, George A. Olah, and co-workers, University of Southern California, Los Angeles, USA, have developed a recyclable ruthenium catalyst supported on Nafion – a resin made from tetrafluoroethylene and perfluorovinyl ethers – that works in water as the solvent. The hydration process with the new catalyst gives high yields with a variety of functionalized nitriles. The catalyst is easy to make by ion exchange and can be recycled several times without losing its catalytic activity.
- Nafion-Ru: A Sustainable Catalyst for Selective Hydration of Nitriles to Amides,
G. K Surya Prakash, Socrates B. Munoz, Attila Papp, Kamil Masood, Inessa Bychinskaya, Thomas Mathew, George A. Olah,
Asian J. Org. Chem. 2012.
DOI: 10.1002/ajoc.201200043