Asexual and Sexual Reproduction
Diatoms (unicellular photosynthetic organisms) reproduce through asexual cell division alternating with short periods of sexual reproduction. A German and Belgian team has now determined that pheromones play an important role in this. As the researchers report in the journal Angewandte Chemie, they were able to isolate and structurally characterize one of these lures.
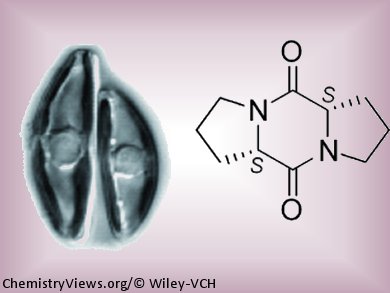
Diatoms have hard mineral shells that are formed like a hatbox from two overlapping halves. During the asexual reproduction phase, the new cells each get one half of the original shell and make the second half themselves. Because the new part of the shell is made inside the shell of the mother cell, the cell size of a given population constantly decreases. Once a critical cell size is reached, the diatoms must switch to sexual reproduction in order to survive. This produces a cell of the original size.
Messanger Molecule for Reproduction
There are indications that for some species, pheromones are involved as regulators in the differentiation and pairing of the cells. The chemical structure of these signaling molecules was previously unknown. A team from the Universities of Ghent, Belgium, and Jena, Germany, has now researched the role of such messenger molecules in the reproduction of Seminavis robusta.
When S. robusta reaches its species-specific critical cell size, two types of sexual cell are differentiated; these are designated as + and – . Afterwards, all of the cells of the mobile + mating type gather around an attracting – cell. A team led by Georg Pohnert, Marnik Vuylsteke, and Wim Vyverman has now been able to prove that both mating types produce chemical signals that activate the mating behavior of their partner. To do this, the researchers extracted the culture medium from diatoms in various states and used this extracted material as a source of pheromones.
They demonstrated that chemical messengers must be activated in order to herald the sexual readiness of the two types of cells. The – cells thus release substances that greatly increase the mobility of the + cells; and the + cells release substances that induce the – cells to become prepared to mate and cause them to excrete the actual attracting pheromone. Both mating types rely on signaling molecules to ensure the presence of a mature mating partner before they invest in a sexual response themselves. This increases the chances of successful sexual reproduction.
Isolating the Lure
By comparing the different metabolic products released during the different phases of the reproductive cycle using a method known as “metabolomics”, the researchers found the lure used by the – cells. They isolated it and identified it as diproline. They were able to synthesize this substance by starting from the amino acid proline and determined its absolute configuration.
Research continues in order to determine the extent to which this new pheromone chemistry can be used to promote the growth of algae in aquaculture or to control undesirable algal biofilms.
- Metabolomics Enabeled Structure Elucidation of a Diatom Sex Pheromone,
Jeroen Gillard, Johannes Frenkel, Valerie Devos, Koen Sabbe, Carsten Paul, Martin Rempt, Dirk Inzé, Georg Pohnert, Marnik Vuylsteke, Wim Vyverman,
Angew. Chem. Int. Ed. 2012.
DOI: 10.1002/anie.201208175