The term “water splitting” refers to the conversion of water to hydrogen and oxygen, a research topic which has received extensive study over the last decade. The motivation behind research in this area lies in its potential to become a future energy source which would reduce society’s dependence on fossil fuels. In order to make such processes sustainable, the conversion of water to hydrogen and oxygen must be achieved with solar energy. This provides an incentive for researchers to develop more efficient materials capable of promoting this transformation.
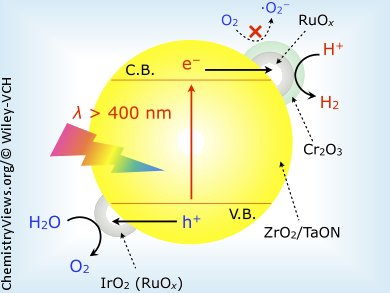
Kazuhiko Maeda and co-workers, Tokyo Institute of Technology, Japan, described a modified tantalum-oxynitride photocatalyst, which is the first example of a d0-type, non-oxide photocatalyst capable of splitting water with only visible light. The TaON was modified with ZrO2 to give a photocatalyst for water reduction. The ZrO2/TaON was then further modified with nanoparticulate cocatalysts, including core/shell-structured RuOx/Cr2O3 nanoparticles and colloidal IrO2.
The IrO2 improved the stability of the water splitting reaction, while the the primary role of RuOx was to extract photogenerated electrons from the conduction band of the TaON component in ZrO2/TaON. The team believe that Cr2O3 modification suppressed photoreduction of O2 on RuOx during the water splitting reaction, thereby allowing for water reduction even in the presence of O2.
- Direct Water Splitting into Hydrogen and Oxygen under Visible Light by using Modified TaON Photocatalysts with d0 Electronic Configuration,
Kazuhiko Maeda, Daling Lu, Kazunari Domen,
Chem. Eur. J. 2013.
DOI: 10.1002/chem.201300158