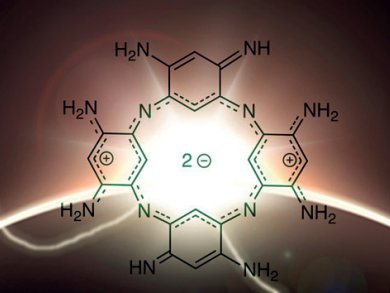
French researchers have synthesized an isostructural porphyrin analogue that does not contain pyrrol subunits. Porphyrins, which are highly conjugated 16-membered macrocycles that contain 18 delocalized π electrons, contain four pyrrol units linked by methine bridges. Denis Jacquemin, Olivier Siri, and their co-workers, Universities of Aix-Marseille and Nantes, have developed a synthetic strategy to form macrocycles that contain four nitrogen atoms separated by three carbon atoms but are “pyrrol-free”.
They started by reacting 1,5-difluoro-2,4-dinitrobenzene with tetraaminobenzene tetrahydrochloride to form a macrocycle, which was then hydrogenated to form the target macrocycle. Nucleus-independent chemical shifts were used to confirm the aromaticity of the compound. An X-ray diffraction study of the protonated form showed that the macrocycle adopts a nonplanar saddle conformation because of the repulsion of the internal C–H hydrogen atoms. These macrocycles are also extremely stable, can form metal complexes, and absorb in the near-infrared region. Thus, the researchers predict that these compounds may have diverse applications.
- Azacalixphyrin: The Hidden Porphyrin Cousin Brought to Light,
Zhongrui Chen, Michel Giorgi, Denis Jacquemin, Mourad Elhabiri, and Olivier Siri,
Angew. Chem. Int. Ed. 2013.
DOI: 10.1002/anie.201301217